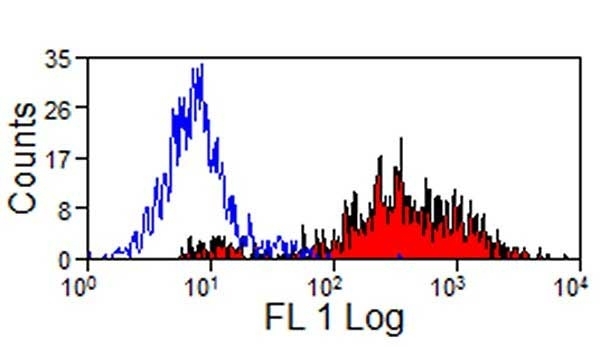
SLA Class II DR antibody | 2E9/13



Mouse anti Pig SLA Class II DR:RPE
- Product Type
- Monoclonal Antibody
- Clone
- 2E9/13
- Isotype
- IgG2b
- Specificity
- SLA Class II DR
| Mouse anti Pig SLA Class II DR antibody, clone 2E9/13 recognizes SLA DR molecules which are expressed on all B cells, antigen presenting cells and on certain subsets of resting and activated T cells. Mouse anti Pig SLA Class II DR antibody, clone 289/13 reacts with lymphocytes from all outbred and miniature pigs so far tested, suggesting that it recognizes a monomorphic determinant of porcine SLA DR. The major histocompatibility complex (MHC) is a cluster of genes that are important in the immune response to infections. In pigs, this is referred to as the swine leukocyte antigen (SLA) region. There are 3 major MHC class II proteins encoded by the SLA which are SLA DP, SLA DQ and SLA DR. Mouse anti pig SLA class II DR, clone 2E9/13 immunoprecipitates a heterodimer composed of two polypeptides of ~28 and ~35 kDa from NP-40 extracts of biotin surface-labeled porcine peripheral blood mononuclear cells. Mouse anti Pig SLA Class II DR antibody, clone 289/13 is reported to inhibit the mixed lymphocyte reaction and T cell stimulation induced by African swine fever virus and staphylococcal enterotoxin B (Bullido et al. 1997). |
- Target Species
- Pig
- Species Cross-Reactivity
-
Target Species Cross Reactivity Bovine - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG conjugated to R. Phycoerythrin (RPE) - lyophilized
- Reconstitution
- Reconstitute with 1 ml distilled water
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
1% bovine serum albumin
5% sucrose - Immunogen
- Porcine monocytes.
- Fusion Partners
- Spleen cells from immunized BALB/c mice were fused with cells of the mouse X63-Ag.8.653 myeloma cell line.
- Max Ex/Em
-
Fluorophore Excitation Max (nm) Emission Max (nm) RPE 488nm laser 496 578 RPE 561nm laser 546 578 - Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
DO NOT FREEZE.
This product should be stored undiluted. This product is photosensitive and should be protected from light.
Avoid repeated freezing and thawing as this may denature the antibody. Should this product contain a precipitate we recommend microcentrifugation before use.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | Neat |
- Flow Cytometry
- Use 10μl of the suggested working dilution to 1x106 cells in 100μl
How to Use the Spectraviewer
Watch the Tool Tutorial Video ▸- Start by selecting the application you are interested in, with the option to select an instrument from the drop down menu or create a customized instrument
- Select the fluorophores or fluorescent proteins you want to include in your panel to check compatibility
- Select the lasers and filters you wish to include
- Select combined or multi-laser view to visualize the spectra
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG2b Negative Control:RPE | MCA691PE | F | 100 Tests | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | Mouse IgG2b Negative Control:RPE | ||||||
References for SLA Class II DR antibody
-
Bullido, R. et al. (1997) Characterization of five monoclonal antibodies specific for swine class II major histocompatibility antigens and crossreactivity studies with leukocytes of domestic animals.
Dev Comp Immunol. 21 (3): 311-22. -
Jeong, H.J. et al. (2010) Comparative measurement of cell-mediated immune responses of swine to the M and N proteins of porcine reproductive and respiratory syndrome virus.
Clin Vaccine Immunol. 17: 503-12. -
Ding, Q. et al. (2011) Human PD-L1-overexpressing porcine vascular endothelial cells induce functionally suppressive human CD4+CD25hiFoxp3+ Treg cells.
J Leukoc Biol. 90 (1): 77-86. -
Thierry, A. et al. (2012) Identification of invariant natural killer T cells in porcine peripheral blood.
Vet Immunol Immunopathol. 149 (3-4): 272-9. -
Iwase H et al. (2015) Initial in vivo experience of pig artery patch transplantation in baboons using mutant MHC (CIITA-DN) pigs.
Transpl Immunol. 32 (2): 99-108. -
Zanotti, C. et al. (2015) Differential Biological Activities of Swine Interferon-α Subtypes.
J Interferon Cytokine Res. 35 (12): 990-1002. -
Wang, Y. et al. (2016) Genipin crosslinking reduced the immunogenicity of xenogeneic decellularized porcine whole-liver matrices through regulation of immune cell proliferation and polarization.
Sci Rep. 6: 24779. -
Gardner, D.S. et al. (2016) Remote effects of acute kidney injury in a porcine model.
Am J Physiol Renal Physiol. 310 (4): F259-71.
View The Latest Product References
-
Singleton, H. et al. (2016) Establishing Porcine Monocyte-Derived Macrophage and Dendritic Cell Systems for Studying the Interaction with PRRSV-1.
Front Microbiol. 7: 832. -
Rahe, M.C. & Murtaugh, M.P. (2017) Interleukin-21 Drives Proliferation and Differentiation of Porcine Memory B Cells into Antibody Secreting Cells.
PLoS One. 12 (1): e0171171. -
Mašek, J. et al. (2017) Multi-layered nanofibrous mucoadhesive films for buccal and sublingual administration of drug-delivery and vaccination nanoparticles - important step towards effective mucosal vaccines.
J Control Release. 249: 183-95. -
Ladowski, J.M. et al. (2018) Swine Leukocyte Antigen Class II Is a Xenoantigen.
Transplantation. 102 (2): 249-54. -
Yang, N. et al. (2018) Reduced antigen presentation capability and modified inflammatory/immunosuppressive cytokine expression of induced monocyte-derived dendritic cells from peripheral blood of piglets infected with porcine circovirus type 2.
Arch Virol. 163 (5): 1231-9. -
López, E. et al. (2019) Identification of very early inflammatory markers in a porcine myocardial infarction model.
BMC Vet Res. 15 (1): 91. -
Liu, S. et al. (2019) Endothelial IL-8 induced by porcine circovirus type 2 affects dendritic cell maturation and antigen-presenting function.
Virol J. 16 (1): 154. -
Radlowski, E.C. et al. (2021) Combination-Feeding Causes Differences in Aspects of Systemic and Mucosal Immune Cell Phenotypes and Functions Compared to Exclusive Sow-Rearing or Formula-Feeding in Piglets.
Nutrients. 13(4):1097. -
Arenal, Á. et al. (2022) Effects of Cardiac Stem Cell on Postinfarction Arrhythmogenic Substrate.
Int J Mol Sci. 23 (24): 16211. -
Franzoni, G. et al. (2022) Analyses of the Impact of Immunosuppressive Cytokines on Porcine Macrophage Responses and Susceptibility to Infection to African Swine Fever Viruses.
Pathogens. 11 (2): 166. -
Haach, V. et al. (2023) A polyvalent virosomal influenza vaccine induces broad cellular and humoral immunity in pigs.
Virol J. 20 (1): 181. -
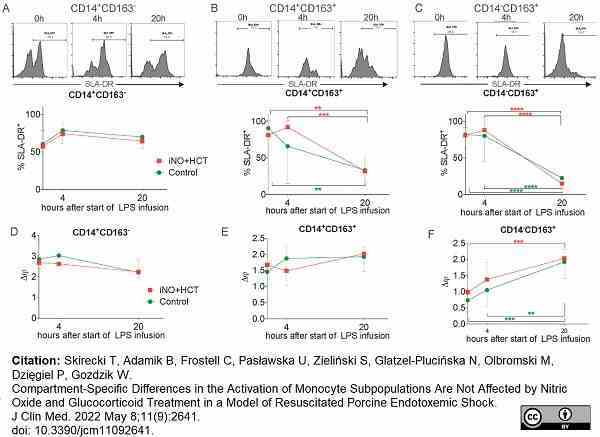
Skirecki, T. et al. (2022) Compartment-Specific Differences in the Activation of Monocyte Subpopulations Are Not Affected by Nitric Oxide and Glucocorticoid Treatment in a Model of Resuscitated Porcine Endotoxemic Shock.
J Clin Med. 11 (9): 2641.
Further Reading
-
Piriou-Guzylack, L. (2008) Membrane markers of the immune cells in swine: an update.
Vet Res. 39: 54. -
Rayat GR et al. (2016) First update of the International Xenotransplantation Association consensus statement on conditions for undertaking clinical trials of porcine islet products in type 1 diabetes - Chapter 3: Porcine islet product manufacturing and release testing criteria.
Xenotransplantation. 23 (1): 38-45.
- UniProt
- Q85ZW4
MCA2314PE
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Pig ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up