Endothelial Cells antibody | MIL11


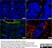
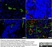
Mouse anti Pig Endothelial Cells
- Product Type
- Monoclonal Antibody
- Clone
- MIL11
- Isotype
- IgE
- Specificity
- Endothelial Cells
| Mouse anti Pig endothelial cells, clone MIL11 raised initially against an immunogen of plastic adherent lymphocytes has been shown to recognize a major subset of porcine endothelial cells. MIL11 was initially identified as staining a characteristic pattern of capillary endothelium in gut forming a network in the lamina propria below the epithelial membrane. Staining was subsequently observed in capillary endothelium in most organs examined, including gut, lung, kidney and skin. Clone MIL11 was observed not to stain endothelium of aorta and other arteries whereas venous endothelium was positive for MIL11 staining. Some population of venous endothelial cells however appear negative for MIL11 staining including those associated with the heart wall, kidney glomeruli and corpus cavernosum (Wilson et al.1996) . |
- Target Species
- Pig
- Product Form
- Tissue culture supernatant - liquid
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
- Immunogen
- Plastic adherent porcine peripheral blood lymphocytes.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Immunohistology - Frozen | 1/20 | 1/100 |
References for Endothelial Cells antibody
-
Wilson, A. D. et al. (1996) Expression of major histocompatibility complex class II antigens, on normal porcine intestinal endothelium.
Immunology 88: 98-103. -
Harrower, T.P. et al. (2006) Long-term survival and integration of porcine expanded neural precursor cell grafts in a rat model of Parkinson's disease.
Exp Neurol.197: 56-69. -
Inman, C.F. et al. (2010) Dendritic cells interact with CD4 T cells in intestinal mucosa.
J Leukoc Biol. 88 (3): 571-8. -
Cho, P.S. et al. (2008) Immunogenicity of umbilical cord tissue derived cells.
Blood. 111 (1): 430-8. -
Inman, C.F. et al. (2010) Rearing environment affects development of the immune system in neonates.
Clin Exp Immunol. 160: 431-9. -
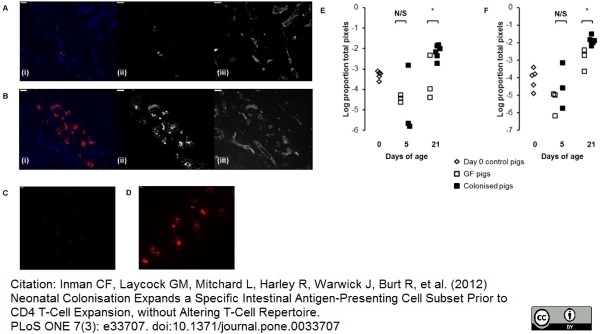
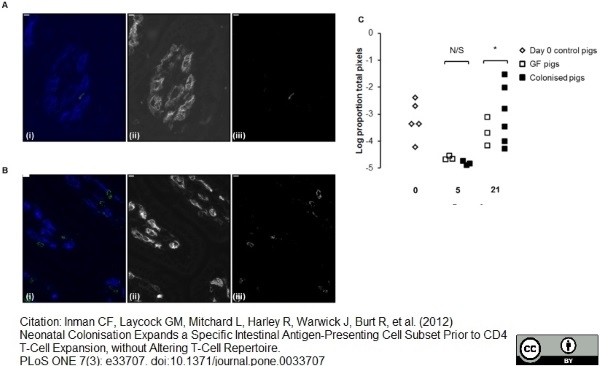
Inman, C.F. et al. (2012) Neonatal colonisation expands a specific intestinal antigen-presenting cell subset prior to CD4 T-cell expansion, without altering T-cell repertoire.
PLoS One. 7: e33707. -
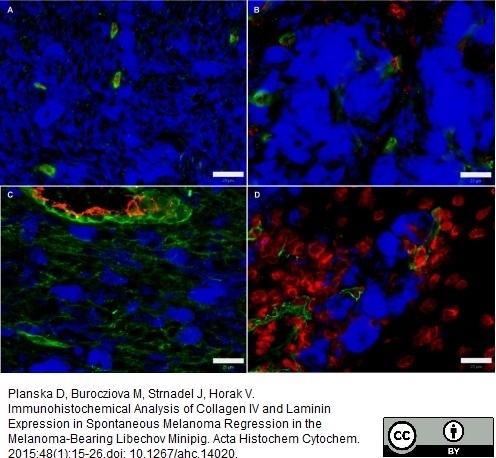
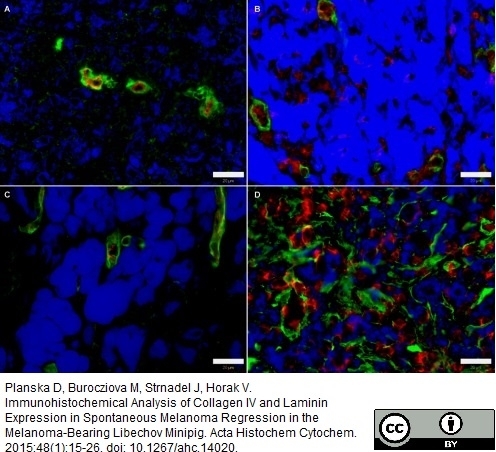
Planska, D. et al. (2015) Immunohistochemical Analysis of Collagen IV and Laminin Expression in Spontaneous Melanoma Regression in the Melanoma-Bearing Libechov Minipig.
Acta Histochem Cytochem. 48 (1): 15-26.
Further Reading
-
Piriou-Guzylack, L. (2008) Membrane markers of the immune cells in swine: an update.
Vet Res. 39: 54.
- RRID
- AB_322717
MCA1752
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Pig ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up