Ovalbumin antibody | 2C6
Mouse anti Ovalbumin
- Product Type
- Monoclonal Antibody
- Clone
- 2C6
- Isotype
- IgE
- Specificity
- Ovalbumin
Filter by Application:
E Reset| Mouse anti Ovalbumin antibody, clone 2C6 recognises ovalbumin (OVA). The antibody is suitable for use as a mouse IgE standard in ELISA assays (Hamadaet al. 2003). |
- Target Species
- Chicken
- Product Form
- Purified IgE - liquid
- Preparation
- Purified IgE prepared from tissue culture supernatant.
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
- Immunogen
- Ovalbumin.
- Approx. Protein Concentrations
- IgE concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunized Balb/c mice were fused with cells of the mouse myeloma, P3U1.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| ELISA | 1/1000 | 1/5000 |
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Rat anti Mouse IgE Heavy Chain:HRP | MCA419P | E | 0.5 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Rat anti Mouse IgE Heavy Chain:HRP | ||||||
Source Reference
-
Hamada, K. et al. (2003) Allergen-independent maternal transmission of asthma susceptibility.
J Immunol. 170: 1683-9.
References for Ovalbumin antibody
-
Suzaki, Y. et al. (2005) A potent antiangiogenic factor, endostatin prevents the development of asthma in a murine model.
J Allergy Clin Immunol. 116 (6): 1220-7. -
Fairley, K.J. et al. (2007) Exposure to the immunosuppressant, perfluorooctanoic acid, enhances the murine IgE and airway hyperreactivity response to ovalbumin.
Toxicol Sci. 97 (2): 375-83. -
Chida, Y. et al. (2007) Early-life psychological stress exacerbates adult mouse asthma via the hypothalamus-pituitary-adrenal axis.
Am J Respir Crit Care Med. 175: 316-22. -
Suzaki, Y. et al. (2007) A small-molecule compound targeting CCR5 and CXCR3 prevents airway hyperresponsiveness and inflammation.
Eur Respir J. 31: 783-9. -
Kambayashi, T. et al. (2008) Indirect involvement of allergen-captured mast cells in antigen presentation.
Blood. 111:1489-96. -
Stevens, T. et al. (2008) Increased transcription of immune and metabolic pathways in naive and allergic mice exposed to diesel exhaust.
Toxicol Sci. 102: 359-70. -
Ellertsen,L.K. et al. (2010) Maternal allergen immunisation to prevent sensitisation in offspring: Th2-polarising adjuvants are more efficient than a Th1-polarising adjuvant in mice.
BMC Immunol. 11: 8-17 -
Paliwal, S. et al. (2010) One-step acquisition of functional biomolecules from tissues.
Proc Natl Acad Sci U S A. 107: 14627-32.
View The Latest Product References
-
Cheung DS et al. (2010) Development of atopy by severe paramyxoviral infection in a mouse model.
Ann Allergy Asthma Immunol. 105 (6): 437-443.e1. -
Piro,.B. et al. (2011) Towards the detection of human papillomavirus infection by a reagentless electrochemical peptide biosensor
Electrochimica Acta. 56 (28): 10688-10693. -
Hansen, J.S. et al. (2011) Determinants of experimental allergic responses: interactions between allergen dose, sex and age.
Scand J Immunol. 73 (6): 554-67. -
Nygaard, U.C. et al. (2015) Early life exposure to bisphenol A investigated in mouse models of airway allergy, food allergy and oral tolerance.
Food Chem Toxicol. 83: 17-25. -
Andreassen, M. et al. (2015) Cry1Ab protein from Bacillus thuringiensis and MON810 cry1Ab-transgenic maize exerts no adjuvant effect after airway exposure.
Scand J Immunol. 81 (3): 192-200. -
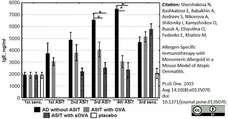
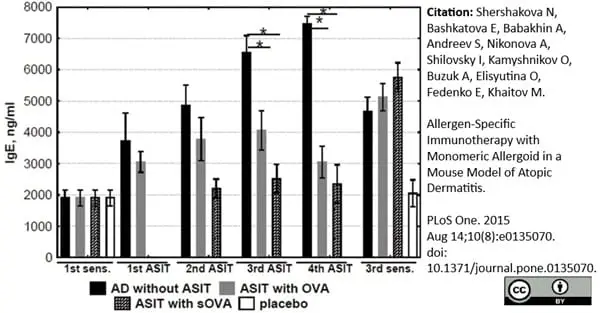
Shershakova N et al. (2015) Allergen-Specific Immunotherapy with Monomeric Allergoid in a Mouse Model of Atopic Dermatitis.
PLoS One. 10 (8): e0135070. -
Diesner, S.C. et al. (2016) A distinct microbiota composition is associated with protection from food allergy in an oral mouse immunization model.
Clin Immunol. 173: 10-18. -
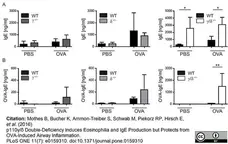
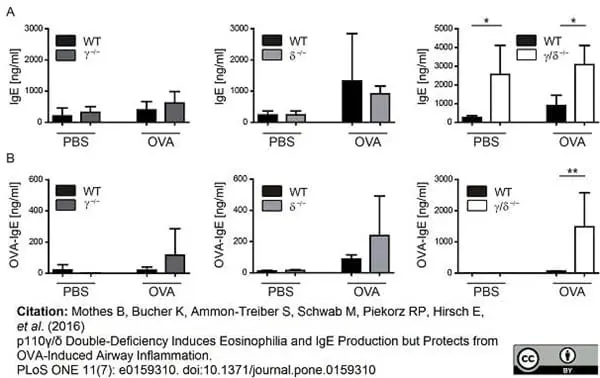
Mothes, B. et al. (2016) p110γ/δ Double-Deficiency Induces Eosinophilia and IgE Production but Protects from OVA-Induced Airway Inflammation.
PLoS One. 11 (7): e0159310. -
Garbani, M. et al. (2017) Allergen-loaded strontium-doped hydroxyapatite spheres improve allergen-specific immunotherapy in mice.
Allergy. 72 (4): 570-8. -
Shin, W.et al. (2018) V-set and Ig domain-containing 4 (VSIG4)-expressing hepatic F4/80+ cells regulate oral antigen-specific responses in mouse.
Eur J Immunol. 48 (4): 632-43. -
Ghonim, M.A. et al. (2018) Sulfated non-anticoagulant heparin blocks Th2-induced asthma by modulating the IL-4/signal transducer and activator of transcription 6/Janus kinase 1 pathway.
J Transl Med. 16 (1): 243. -
Haselmayer, P. et al. (2019) Efficacy and Pharmacodynamic Modeling of the BTK Inhibitor Evobrutinib in Autoimmune Disease Models.
J Immunol. 202 (10): 2888-906. -
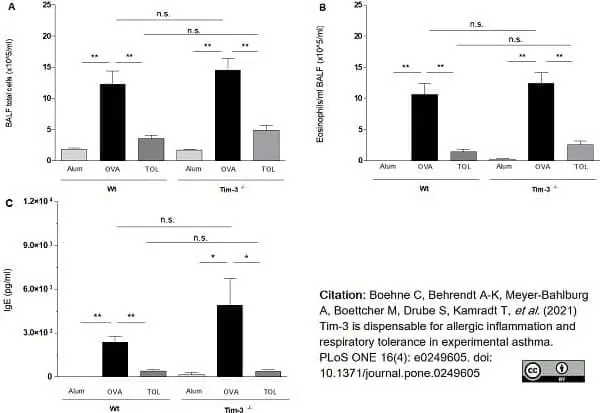
Boehne, C. et al. (2021) Tim-3 is dispensable for allergic inflammation and respiratory tolerance in experimental asthma.
PLoS One. 16 (4): e0249605. -
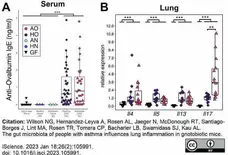
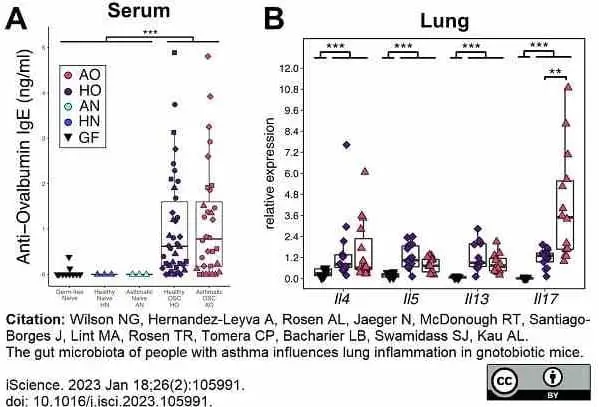
Wilson, N.G. et al. (2023) The gut microbiota of people with asthma influences lung inflammation in gnotobiotic mice.
iScience. 26 (2): 105991. -
Gnipp, S. et al. (2018) Nitric oxide dependent signaling via cyclic GMP in dendritic cells regulates migration and T-cell polarization.
Sci Rep. 8 (1): 10969. -
Tsai, C.T. et al. (2018) Isotype-specific agglutination-PCR (ISAP): A sensitive and multiplex method for measuring allergen-specific IgE.
J Allergy Clin Immunol. 141 (5): 1901-4.e15. -
Hjálmsdóttir, Á. et al. (2024) T cell independent antibody responses with class switch and memory using peptides anchored on liposomes.
NPJ Vaccines. 9 (1): 115.
Patent
-
Mitragotri, S. et al. (2015) Compositions for Solubilizing Cells and/or Tissue
Pat app Pub: US 2015/0275174 A1
- RRID
- AB_2285753
MCA2259
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Chicken ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up