Bu-1A antibody | L22



Mouse anti Chicken Bu-1A:FITC
- Product Type
- Monoclonal Antibody
- Clone
- L22
- Isotype
- IgG1
- Specificity
- Bu-1A
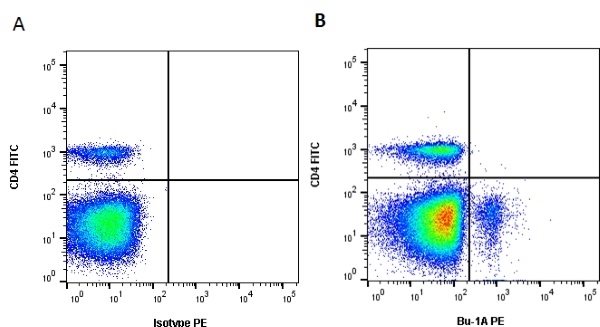
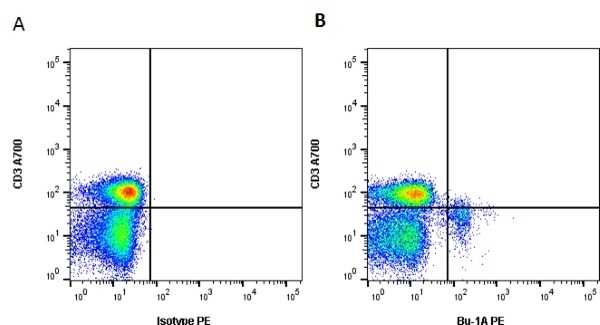
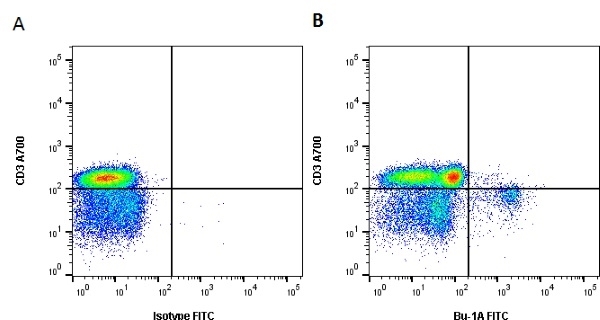
| Mouse anti Chicken Bu-1a, clone L22 recognises the 'a' allele of the polymorphic cell surface antigen Bu-1, also known as chB6. Bu-1a is a 70 kDa alloantigen expressed on B cells and a subset of macrophages. Bu-1 is expressed on B cell precursors at early stages of development (Houssaint et al. 1991). Weak cross-reactivity with Bu-1b may be seen at higher antibody levels. This may be removed by appropriate titration. Mouse anti chicken BU-1a, clone L22 cross reacts with quail Bu-1 but does not appear reactive with either turkey or guinea fowl tissues (Igyarto et al. 2008) |
- Target Species
- Chicken
- Species Cross-Reactivity
-
Target Species Cross Reactivity Quail Turkey Guinea Fowl - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG conjugated to Fluorescein Isothiocyanate Isomer 1 (FITC) - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
1% bovine serum albumin - Immunogen
- Chicken Bursa cells.
- Approx. Protein Concentrations
- IgG concentration 0.1 mg/ml
- Fusion Partners
- Spleen cells from immunised Balb/c mice were fused with cells of the mouse SP2/0-Ag14 myeloma cell line.
- Max Ex/Em
-
Fluorophore Excitation Max (nm) Emission Max (nm) FITC 490 525 - Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended. This product is photosensitive and should be protected from light.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/20 | 1/80 |
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 106 cells in 100μl
Source Reference
-
Pink, J.R. & Rijnbeek, A.M. (1983) Monoclonal antibodies against chicken lymphocyte surface antigens.
Hybridoma. 2: 287-96.
References for Bu-1A antibody
-
Chaussé, A.M. et al. (2011) Expression of toll-like receptor 4 and downstream effectors in selected cecal cell subpopulations of chicks resistant or susceptible to salmonella carrier state.
Infect Immun. 79: 3445-54. -
Houssaint, E. et al. (1989) Bu-1 antigen expression as a marker for B cell precursors in chicken embryos.
Eur J Immunol. 19 (2): 239-43. -
Tregaskes, C.A. et al. (1996) Chicken B-cell marker chB6 (Bu-1) is a highly glycosylated protein of unique structure.
Immunogenetics. 44: 212-7. -
Houssaint, E. et al. (1991) Early separation of B and T lymphocyte precursors in chick embryo.
J Exp Med. 174: 397-406. -
Igyártó, B.Z. et al. (2008) Identification of the avian B-cell-specific Bu-1 alloantigen by a novel monoclonal antibody.
Poult Sci. 87: 351-5. -
Veromaa, T. et al. (1988) Monoclonal antibodies against chicken Bu-1a and Bu-1b alloantigens.
Hybridoma. 7: 41-8. -
Chaussé, A.M. et al. (2014) Susceptibility to Salmonella carrier-state: a possible Th2 response in susceptible chicks.
Vet Immunol Immunopathol. 159: 16-28. -
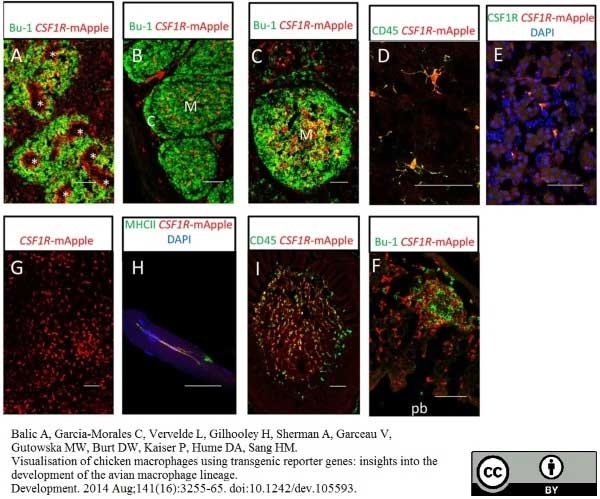
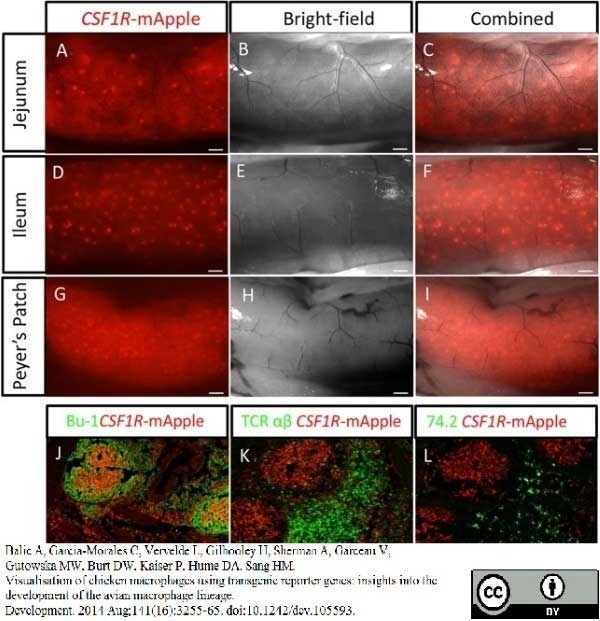
Balic, A. et al. (2014) Visualisation of chicken macrophages using transgenic reporter genes: insights into the development of the avian macrophage lineage.
Development. 141: 3255-65.
View The Latest Product References
-
Luna-Acosta, J.L. et al. (2015) Direct antiapoptotic effects of growth hormone are mediated by PI3K/Akt pathway in the chicken bursa of Fabricius.
Gen Comp Endocrinol. 224: 148-59. -
Jarosz, Ł. et al. (2017) The effect of feed supplementation with zinc chelate and zinc sulphate on selected humoral and cell-mediated immune parameters and cytokine concentration in broiler chickens.
Res Vet Sci. 112: 59-65. -
Smialek, M. et al. (2017) Immunological aspects of the efficiency of protectotype vaccination strategy against chicken infectious bronchitis.
BMC Vet Res. 13 (1): 44. -
Jarosz, Ł.S et al. (2018) The effect of feed supplementation with a copper-glycine chelate and copper sulphate on selected humoral and cell-mediated immune parameters, plasma superoxide dismutase activity, ceruloplasmin and cytokine concentration in broiler chickens.
J Anim Physiol Anim Nutr (Berl). 102 (1): e326-e36.
- Synonyms
- Bursal Antigen 1 A
- RRID
- AB_2234729
- UniProt
- Q90746
MCA2170F
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Chicken ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up