Mumps Virus


Native Mumps Virus
- Product Type
- Antigen
- Specificity
- Mumps Virus
| Native mumps virus preparation Native mumps virus contains a high concentration of virus and viral components. The preparation also contains some cellular material suspended in tissue culture medium (1969B). Mumps virus is a member of the Paramyxoviridae family and is the causative agent of mumps. Mumps is most typically characterised by painful swelling of the salivary glands and fever. In some adult men, the virus might also infect the testicles, rarely causing sterility. The virus is spread by airborne droplets from the nose or throat and can have an incubation period of up to three weeks. Infected individuals are contagious from approximately 6 days before until about 9 days after the onset of symptoms. The disease is self-limiting and treatment involves controlling specific symptoms. An effective vaccine exists and may be given separately or as part of the MMR vaccine. |
- Target Species
- Viral
- Product Form
- Inactivated Mumps virus - liquid
- Preparation
- Mumps virus, Enders strain, cultured in BSC-1 cells. Optimally infected cells are harvested, disrupted by sonication in culture medium and subjected to low speed centrifugation. The supernatant from the infected culture is concentrated by crossflow ultrafiltration. The antigen preparation is inactivated using gamma radiation, which primarily damages viral genetic material.
- Buffer Solution
- Culture medium 1969B
- Preservative Stabilisers
- None present
- Activity
- Antigenic activity is 51.1% of internal reference batch
- Approx. Protein Concentrations
- Current, batch-specific concentration 1.0 mg/ml
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Storage in frost-free freezers is not recommended.
This product should be stored undiluted. Avoid repeated freezing and thawing as this may denature the protein.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| ELISA |
- Instructions For Use
- PIP014 should be sonicated immediately before use to ensure the preparation is uniform. The product may be used in a variety of immunoassay formats or may be further purified to meet the requirements of a particular assay format.
References for Mumps Virus
-
Bhoj, V.G. et al. (2016) Persistence of long-lived plasma cells and humoral immunity in individuals responding to CD19-directed CAR T-cell therapy.
Blood. 128 (3): 360-70. -
Riggenbach, M.M. et al. (2021) Mumps virus-specific immune response outcomes and sex-based differences in a cohort of healthy adolescents.
Clin Immunol. 234: 108912. -
Böröcz, K. et al. (2022) Dynamic Features of Herd Immunity: Similarities in Age-Specific Anti-Measles Seroprevalence Data between Two Countries of Different Epidemiological History.
J Clin Med. 11(4):1145. -
Wang, L. et al. (2023) Associations of adaptive immune cell subsets with measles, mumps, and Rubella−Specific immune response outcomes
Heliyon. 9 (12): e22998. -
Ovsyannikova, I.G. et al. (2023) Genome-wide determinants of cellular immune responses to mumps vaccine.
Vaccine. 41 (44): 6579-88. -
Szinger, D. et al. (2023) Following Natural Autoantibodies: Further Immunoserological Evidence Regarding Their Silent Plasticity and Engagement in Immune Activation.
Int J Mol Sci. 24 (19)Oct 06 [Epub ahead of print]. -
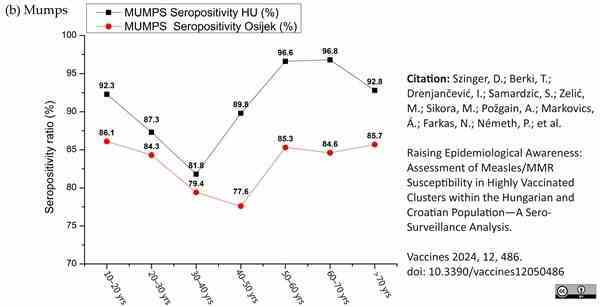
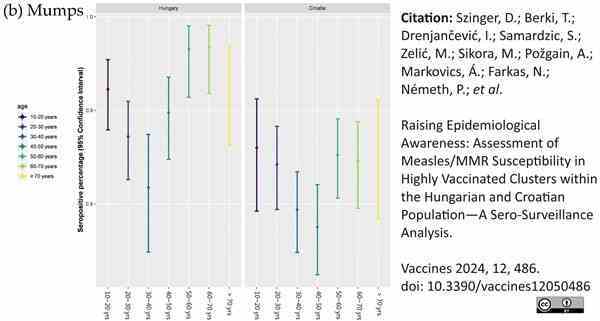
Szinger, D. et al. (2024) Raising Epidemiological Awareness: Assessment of Measles/MMR Susceptibility in Highly Vaccinated Clusters within the Hungarian and Croatian Population—A Sero-Surveillance Analysis
Vaccines. 12 (5): 486.
- UniProt
- P21277
- GO Terms
- GO:0003723 RNA binding
- GO:0030529 ribonucleoprotein complex
- GO:0005198 structural molecule activity
- GO:0019013 viral nucleocapsid
- GO:0030430 host cell cytoplasm
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Viral ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up