
Popular topics

-
References
Kee N et al. (2002). The utility of Ki-67 and BrdU as proliferative markers of adult neurogenesis. J Neurosci Methods 115, 97-105.
Top Tips for Troubleshooting Your BrdU Blues
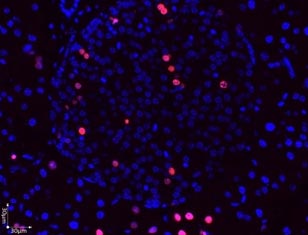
Accurate measurement of cell division and proliferation is a fundamental part of experimental biology and is crucial in the assessment of cell health. Various methods can be used to detect cell division like:
- Continuous visualization of cells in culture via microscopy
- Dilution of vital dyes
- Immunodetection of proliferation markers like PCNA and Ki67
- Use of non-radioactive thymidine analogs, such as 5-bromodeoxyuridine (BrdU)
This blog post focuses on BrdU and our top tips and tricks for conducting a BrdU labeling experiment. This should help you to plan your BrdU experiments and troubleshoot any BrdU blues you encounter.
Measuring Proliferation with Thymidine Analogs
Thymidine analogs like BrdU, chloro-deoxyuridine (CldU), iodo-deoxyuridine (IdU), and 5-ethynyl-2′-deoxyuridine (EDU) have structures that are highly similar to thymidine and they can be specifically incorporated into DNA during the S phase of the cell cycle instead of thymidine. In the case of BrdU, bromine replaces the methyl group at carbon 5 of the thymine portion (Figure 1).
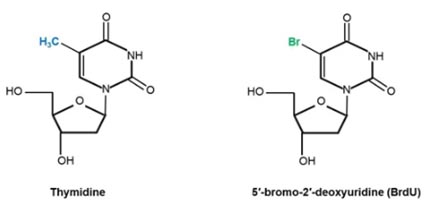
Fig.1. Chemical structures of the DNA nucleoside thymidine and its analog BrdU. In the case of BrdU, the 5-methyl group (shown in blue) of thymidine has been substituted by bromine (highlighted in green).
This structural change, although slight enough to enable incorporation into DNA, is significant enough to facilitate detection by antibodies like Mouse Anti-BrdU Antibody, clone Bu20a (MCA2483) and Rabbit Anti-BrdU Antibody (AHP2405). There are several commonly used antibody-based BrdU detection methods including flow cytometry, immunocytochemistry, and immunohistochemistry.
BrdU can be used for tracing cell lineage and cell survival as the label is passed into daughter cells (Figure 2) and retained long term. In fact, BrdU labeling experiments contributed to the current acceptance of neurogenesis as a natural process in mammalian adult brains (Kee et al. 2002).
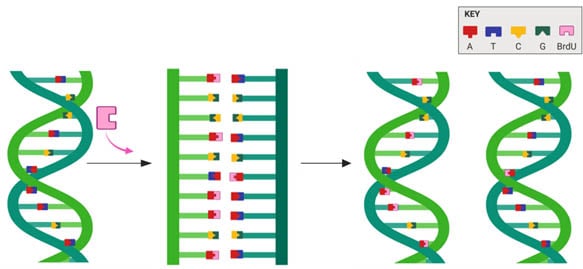
Fig.2. BrdU is incorporated into newly synthesized DNA in place of thymidine.
Top Tips for BrdU Labeling Experiments
BrdU labeling experiments can provide detailed information about cell proliferation within a sample, but this is only possible with properly designed experiments and appropriate controls. Here are some best practices that you can adopt to get great results.
1. Titrate to Find Optimal BrdU and Antibody Concentration
The concentration of BrdU and incubation time required for optimal labeling depends on how rapidly the cells divide and this will need to be optimized for the cells or tissue you are using. Perform titration experiments to determine the concentration of BrdU that will give the best level of incorporation, without inducing cytotoxicity in your cells. You should also perform titration experiments in order to determine the optimal concentration of your anti-BrdU antibody.
2. Include Controls in Your Set-up
Your results are meaningless without proper controls! You should make sure that all the relevant controls are included. These could be:
- Negative control cells: treat your cells with only the solvent that the stock BrdU solution is diluted in, to observe any effect that the diluent used to prepare the stock solution has on your cells. For example, if your stock BrdU solution is diluted in dimethyl sulfoxide (DMSO), you should prepare some negative control cells which are treated with DMSO only. It is essential that volume of diluent you administer to your negative control group is the same as the BrdU administered to your treated cells, animals or tissue and that the incubation period is the same for both sets of cells
- Secondary only antibody controls: if you are using secondary antibodies, it is advisable to run samples that are assayed with just the secondary antibody (without using the primary BrdU antibody) to check for background staining that results from nonspecific binding of the secondary antibody
- Isotype controls: consider whether you should include isotype controls. This involves staining a sample of your cells or tissue with an antibody that binds to an unrelated target (that is one that is not designed to target BrdU) but is of the same isotype as the antibody targeting BrdU
- Positive controls: cells, or tissue slides, can be helpful to enable you to check whether your anti-BrdU antibody and staining procedure are suitable for BrdU. Include a nuclear or DNA dye as a counterstain, like PureBlu DAPI (1351303), to the antibody targeting BrdU in order to determine the specificity of any positive staining observed. The DNA dye selected should have different excitation and emission spectra to the fluorophore used to detect your anti-BrdU antibody
3. Select the Right Antibody
One of the most important stages when planning any experiment is antibody selection. When choosing an anti-BrdU antibody for your experiment, look for an antibody which has a different host species to the cells or tissue you plan to use for your experiment. This antibody should also have supporting data that shows characteristic positive staining of BrdU in your chosen application, which can usually be found on the datasheet or product webpage.
If you are looking to perform a dual pulse labeling experiment, select an anti-BrdU antibody that has been tested for cross-reactivity with thymidine and other thymidine analogs.
4. Optimize the Protocol for Your Chosen Method for Immunodetection of BrdU
Keep in mind that the washing and blocking buffers you use may also need to be optimized for the anti-BrdU antibody you are using in order to give the best signal to background ratio from your chosen anti-BrdU antibody. The DNA denaturation step is required to allow the anti-BrdU antibody access to the BrdU incorporated into your cells and should also be optimized. Establish the concentration of hydrochloric acid, temperature, and incubation period that enables BrdU detection, while keeping the structure of proteins in the cells intact. This step is particularly important if you wish to counterstain BrdU with an antibody targeting a cytoplasmic marker such as GAPDH, or cell proliferation markers like Ki67 or PCNA.
I hope that by bearing in mind these few basic tips when planning your BrdU labeling experiments, you will be able to have confidence in the results you obtain and successfully troubleshoot most of the unexpected results that you encounter.
Webinar
This webinar provides a guide to best practices for performing BrdU labeling experiments and subsequent detection in immunoassays, helping you to avoid BrdU blues.
BRDU Staining Tips
For more useful advice, including a selection of handy pdfs, why not take a look at our main Brdu Staining Tips page.
References
Kee N et al. (2002). The utility of Ki-67 and BrdU as proliferative markers of adult neurogenesis. J Neurosci Methods 115, 97-105.
You may also be interested in...

View more Tips&Tricks or Applications blogs















