
Popular topics

-
References
Altman J and Das GD (1965). Autoradiographic and histological evidence of postnatal hippocampal neurogenesis in rats. J Comp Neurol 124, 319-335.
Eriksson PS et al. (1998). Neurogenesis in the adult human hippocampus. Nat Med 4, 1313-1317.
Gratzner HG (1982). Monoclonal antibody to 5-bromo-and 5-iododeoxyuridine: a new reagent for detection of DNA replication. Science 218, 474-475.
Iball J et al. (1966). Structures of 5-bromodeoxyuridine and 5-bromouridine. Nature 209, 1230-1232.
Kolb B et al. (1999). Embryonic and postnatal injections of bromodeoxyuridine produce age-dependent morphological and behavioral abnormalities. J Neuro 19, 2337-2346.
Lehner B et al. (2011). The dark side of BrdU in neural stem cell biology: detrimental effects on cell cycle, differentiation and survival. Cell Tissue Res 345, 313-328.
National Center for Biotechnology Information. PubChem Compound Database; CID=6035, https://pubchem.ncbi.nlm.nih.gov/compound/6035 (accessed March 17, 2017).
National Center for Biotechnology Information. PubChem Compound Database; CID=5789, https://pubchem.ncbi.nlm.nih.gov/compound/5789 (accessed March 17, 2017).
Waldman F et al. (1988). Clinical applications of the bromodeoxyuridine/DNA assay. Cytometry Supplement 3, 65-72.
Young DW et al. (1969). The crystal and molecular structure of thymidine. Aca Cryst B25, 1423-1432.
Bromodeoxyuridine (BrdU) in adult neurogenesis research - Part 1
Up until the mid-1960s, scientists believed that the generation of functional neurons from neural precursors (neurogenesis) only occurred during the embryonic and peri-natal stages in mammals. Essentially, it was thought that loss of neurons was an irreversible process in the adult brain. Altman and Das (1965) provided the first evidence of adult neurogenesis in post-natal rats using autoradiographic analysis of [3H] thymidine incorporation into DNA during the S phase of cell growth. However, it was still unclear whether new neurons were produced in the brains of adult humans.
By the 1990s, scientists had begun studying DNA replication using non-autoradiography techniques. Most predominant was the use of bromodeoxyuridine (BrdU), an artificial nucleoside that substitutes for thymine during DNA replication. Although it is a thymidine analog, BrdU is a halopyrimidine with a different molecular structure than thymidine (see image below). It allows faster detection of DNA synthesis compared to use of [3H] thymidine. In addition, unlike labeled thymidine, it can easily be detected by immunohistochemical methods, which also allow the design of multiplex IHC experiments by simultaneously staining for neuronal stem cell markers such as Nestin and SOX2 (Gratzner 1982).

Prefer to listen to the BrdU in adult neurogenesis research story?
Check out the podcast version of our two blog articles. You will learn how BrdU was established as a popular tool for neurogenesis research as well as gain insight into the latest BrdU related findings. Finally, you will go away with 5 practical tips for controlling your BrdU labeling experiments. Spend your experimental break wisely and get some science on the go in less than 15 minutes.
Duration: 12:23 | File size: 5560KB | Recommended browsers: Google Chrome and Mozilla Firefox.
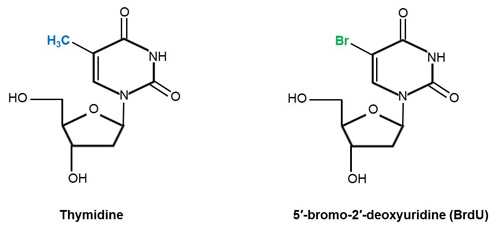
Chemical structures of thymidine and its analog 5′-bromo-2′-deoxyuridine (BrdU). The two structures are identical apart from the fact that the methyl group (highlighted in blue) on carbon residue 5 of thymidine (PubChem CID: 5789) has been substituted by bromine (highlighted in green) (Kolb et al. 1999). Addition of BrdU (PubChem CID: 6035) to cells introduces a bromine atom (Br) into the DNA of cells in the S phase of the cell cycle (Iball et al. 1966, Young et al. 1969). Structures adapted from Iball et al. (1966) (BrdU) and Young et al. (1969) (thymidine).
In clinical settings, BrdU has been used to monitor cancer progression (Waldman et al. 1988). To evaluate tumor growth, BrdU is injected into a patient’s bloodstream. A biopsy of the tumor is then taken and an anti-BrdU antibody is used to detect cells that have incorporated BrdU into their DNA. The ratio of BrdU positive cells to total tumor cells gives an indication of the rate of cell growth.
Neuroscientist Professor Fred Gage of Salk Research Institute for Biological Sciences later adapted this application of BrdU to neuroscience by hypothesizing that BrdU could be used to birthdate individual cells; he reasoned that the chemical would be incorporated into new but not old brain cells (Eriksson et al. 1998). He further speculated that if BrdU was incorporated into the tumor after injection into the bloodstream, the chemical was also likely incorporated into brain cells. To test their hypothesis Prof. Gage and his research team acquired post-mortem brain samples from cancer patients administered BrdU and set out to answer the question of whether neurogenesis occurs in adult human brains.
Using immunofluorescent labeling of BrdU and the neuronal markers NeuN, calbindin and neuron specific enolase (NSE), Prof. Gage and his team determined that new neurons were generated from dividing progenitor cells in the dentate gyrus of adult humans (Eriksson et al. 1998). They also demonstrated that the human hippocampus retains its ability to generate neurons throughout life (Eriksson et al. 1998).
These findings overturned the long standing dogma that neurogenesis did not occur in adults. The neurogenesis field subsequently took off, and BrdU is now routinely used in neurogenesis studies as a lineage tracer. As a result, BrdU has contributed significantly to our understanding of the timing and pattern of neurogenesis. It is currently the research tool of choice for neurogenesis studies; however, recent studies indicate off-target effects that should be carefully controlled for in your experimental design (Lehner et al. 2011).
Stay tuned to our next blog post where we discuss the “dark side of BrdU” and how you can carefully control your BrdU experiments for accurate data analysis (Lehner et al. 2011).
In the meantime, check out our mouse anti-BrdU antibody (clone Bu20a), which has been used in several neuroscience publications.
Application of mouse anti-BrdU antibody, clone Bu20a (MCA2483) in neurogenesis studies
- Kent BA et al. (2015). The orexigenic hormone acyl-ghrelin increases adult hippocampal neurogenesis and enhances pattern separation. Psychoneuroendocrinology 51, 431-439.
- Laitman BM et al. (2016). The transcriptional activator Krüppel-like factor-6 is required for CNS myelination. PLoS Biol 14, e1002467.
- Pappalardo LW et al. (2014). Voltage-gated sodium channel Nav 1.5 contributes to astrogliosis in an in vitro model of glial injury via reverse Na+ /Ca2+ exchange. Glia 62, 1162-1175.
- Sato Y et al. (2013). Grafting of neural stem and progenitor cells to the hippocampus of young, irradiated mice causes gliosis and disrupts the granule cell layer. Cell Death Dis 4, e591.
- Xie LL et al. (2009). Aquaporin 4 knockout resists negative regulation of neural cell proliferation by cocaine in mouse hippocampus. Int J Neuropsychopharmacol 12, 843-850.
In addition to its wide application in neuroscience, the antibody is also suitable for monitoring BrdU incorporation by flow cytometric analysis.
In the image below, we analyzed human BrdU-labeled lymphoma cells using our mouse anti-BrdU antibody, clone Bu20a (MCA2483). The image shows cell cycle profiles, BrdU positive cells are in the S-phase of the cell cycle. For more information about studying the incorporation of BrdU by flow cytometry, check out the respective chapter in our Flow Cytometry Basics Guide.
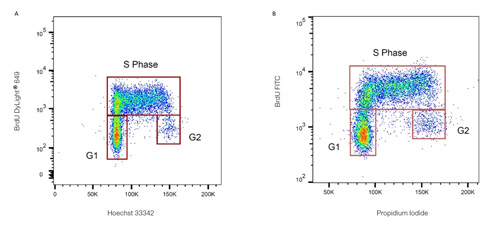
Flow cytometry analysis of human BrdU-labeled lymphoma cells stained with mouse anti-BrdU antibody, clone Bu20a. PureBlu™ Hoechst 333342 (A) and ReadiDrop™ Propidium Iodide (B) were used to stain total DNA.
References
Altman J and Das GD (1965). Autoradiographic and histological evidence of postnatal hippocampal neurogenesis in rats. J Comp Neurol 124, 319-335.
Eriksson PS et al. (1998). Neurogenesis in the adult human hippocampus. Nat Med 4, 1313-1317.
Gratzner HG (1982). Monoclonal antibody to 5-bromo-and 5-iododeoxyuridine: a new reagent for detection of DNA replication. Science 218, 474-475.
Iball J et al. (1966). Structures of 5-bromodeoxyuridine and 5-bromouridine. Nature 209, 1230-1232.
Kolb B et al. (1999). Embryonic and postnatal injections of bromodeoxyuridine produce age-dependent morphological and behavioral abnormalities. J Neuro 19, 2337-2346.
Lehner B et al. (2011). The dark side of BrdU in neural stem cell biology: detrimental effects on cell cycle, differentiation and survival. Cell Tissue Res 345, 313-328.
National Center for Biotechnology Information. PubChem Compound Database; CID=6035, https://pubchem.ncbi.nlm.nih.gov/compound/6035 (accessed March 17, 2017).
National Center for Biotechnology Information. PubChem Compound Database; CID=5789, https://pubchem.ncbi.nlm.nih.gov/compound/5789 (accessed March 17, 2017).
Waldman F et al. (1988). Clinical applications of the bromodeoxyuridine/DNA assay. Cytometry Supplement 3, 65-72.
Young DW et al. (1969). The crystal and molecular structure of thymidine. Aca Cryst B25, 1423-1432.
You may also be interested in...

View more cell-cycle or tips-and-tricks blogs














