
Popular topics

-
References
Baker M (2015). Reproducibility crisis: blame it on the antibodies. Nature 521, 274–276.
Baker M (2020). When antibodies mislead: the quest for validation. Nature 585, 313–314.
Fonseca SG et al. (2015). Retraction Note: Wolfram syndrome 1 and adenylyl cyclase 8 interact at the plasma membrane to regulate insulin production and secretion. Nat Cell Bio 17, 105.
Lyst MJ et al. (2015). Retraction: Regulation of MBD1‐mediated transcriptional repression by SUMO and PIAS proteins. EMBO J 38, e103220.
Vasilevsky NA et al. (2013). On the reproducibility of science: unique identification of research resources in the biomedical literature. PeerJ 1, e148.
Uhlen M et al. (2016). A proposal for validation of antibodies. Nat Methods 13, 823–827.
Weller MG (2018). Ten basic rules of antibody validation. Anal Chem Insights, 13, 1177390118757462.
The Importance of Antibody Validation
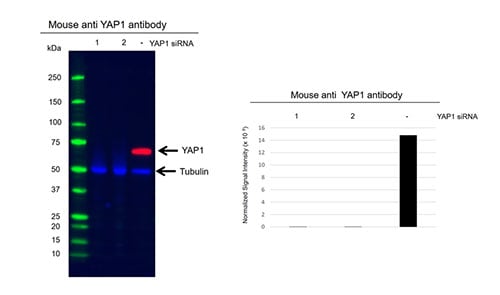
While you might think that the onus on validating antibodies falls solely to antibody suppliers, validating that an antibody works as intended under your own experimental conditions is an important part of the research process. Robust antibody validation ensures that your data can be interpreted correctly and is increasingly asked for by journals, like Nature, when it comes to publication of your findings, to try and tackle the problem of irreproducible data. In this blog, we discuss the importance of antibody validation and highlight steps you can take to ensure you generate publication-quality data.
The Reproducibility Crisis
Antibody validation is the experimental proof that an antibody is suitable for its intended purpose (Weller 2018). That is, the antibody specifically detects only the antigen of interest, works in the desired application, and has consistent performance meaning that results are reproducible.
A lack of reproducibility of vast numbers of well-cited studies has led to a “reproducibility crisis” and in many cases poorly validated antibodies are to blame (Baker 2015). The consequences of not validating an antibody can potentially be catastrophic and compromise your scientific progress, or career. Antibodies that haven’t been thoroughly validated can give false results and lead to retractions of papers (for example Fonseca et al. 2015, Lyst et al. 2015), causing embarrassment to the research group who published the study, as well as grave implications for any research based on the retracted data.
Nonspecific binding, variability between batches, different experimental conditions (like buffer choice, incubation time, or incubation temperature), use in an unintended application, and poor storage conditions can all lead to antibodies giving inconsistent results. Poor antibody choice, or inappropriate use of antibodies, can waste hours of research and a significant proportion of research budgets. End users validating antibodies for their assays, as well as vendors increasing their validation efforts, are helping to tackle the reproducibility crisis.
A number of bodies (like the European Monoclonal Antibodies Network and International Working Group for Antibody Validation) are now issuing recommendations to help you select effective antibodies and guidelines on antibody validation for research (Uhlen et al. 2016).
Think About the Application
Not every antibody can be used in all applications and performance in one application is not indicative of performance in a different one. This is because the target protein may be presented differently depending on the choice of application, such as in a native or denatured conformation, and the antibody may only recognize one particular form. This means that an antibody that is stated to work in immunoprecipitation may not detect the same protein under reducing western blot conditions.
Bio-Rad highlights the tested and recommended applications on the Bio-Rad antibodies website search results pages, product pages, and on product datasheets. We guarantee the performance of the antibody in these stated applications, as described on the datasheet.
Confirm Correct Identification of Your Target
A crucial part of antibody validation is showing that the antibody specifically detects the antigen of interest and there is no cross-reactivity. Detection of a different protein and/or cross-reactivity lead to incorrect observations and assumptions about biological behavior, so it is essential you are certain that your antibody is specific for your target. It is possible to confirm this by validating the antibody yourself (for example using a different antibody to the same target to confirm the result).
Best practice is to always validate an antibody you purchase under your own experimental conditions, as well as to choose a validated antibody like Bio-Rad’s PrecisionAb Antibodies, which include transparent data on performance and detailed information on the conditions it has been tested in.
As an example, for western blots, this means that your antibody gives clean bands of the correct molecular weight and that all appropriate controls are used to eliminate sources of error. Knockout (KO) technology is an ideal way to validate antibody specificity as you can confirm an absence of bands in lysates from KO cells, and a presence of a band of the correct molecular weight in lysates from cell lines where your target is expressed (Figure 1).
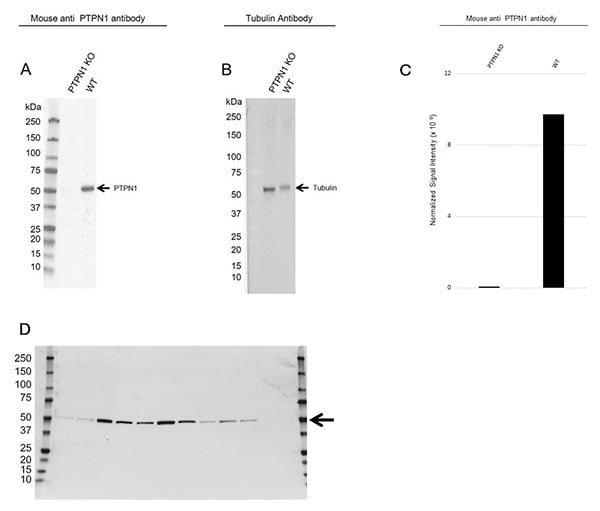
Fig. 1. Validation data for Mouse Anti-PTPN1 Antibody, clone OTI1B4 (VMA00185). A, PTPN1 knockout (KO) HeLa and wild type HeLa (WT) whole cell lysates probed with Mouse Anti-PTPN1 Antibody (VMA00185) followed by detection with HRP conjugated Goat Anti-Mouse IgG (1:10,000 STAR207P) and visualized on the ChemiDoc MP Imaging System with a 210 sec exposure. B, PTPN1 KO HeLa and WT HeLa whole cell lysates probed with hFAB Rhodamine Anti-Tubulin Primary Antibody (12004166) and visualized on the ChemiDoc MP Imaging System with a 30 sec exposure. C, normalized signal intensity of PTPN1 from CRISPR KO and WT lysates. Signal intensity was normalized to the tubulin signal. D, western blot analysis of whole cell lysates probed with Mouse Anti-Human PTPN1 Antibody, clone OTI1B4 (VMA00185) followed by detection with HRP conjugated Goat Anti-Mouse IgG (1/10,000, STAR207P) and visualized on the ChemiDoc MP Imaging System with 7 sec exposure. Arrow points to PTPN1 (molecular weight 50 kDa).
Don’t Base Antibody Selection Solely on Citations
While it is commonplace to choose a highly cited antibody, when looking for an antibody to use in your own studies, it is prudent to look beyond the number of citations and evaluate the quality of the published data. Examine protein expression data and read reviews to identify well-characterized antibodies that give consistent results. Ideally, look for an antibody that has been validated in the same species, tissue type, and technique that you are planning to use.
There are situations where highly cited antibodies may not always be the most appropriate choice. Newer antibodies may have superior performance or work in a greater number of applications, offering you experimental flexibility. There have also been instances where highly cited antibodies have been found later to not behave as expected, meaning assumptions based on their results are incorrect (Baker 2020).
Are you sure that you are choosing the same antibody used in the publications? Ideally, scientists include detailed information about their antibody choice (including host species, vendor, catalog number, and clone name for monoclonal antibodies). However, this is not always the case and antibodies are frequently cited with incomplete information, making it hard to identify the correct product (Vasilevsky et al. 2013). Furthermore, as not all journals require information on antibody validation to be submitted, there may be only limited, or even no information for you to evaluate whether the antibody will work in your experiments.
What Steps Can You Take to Help Tackle the Reproducibility Crisis?
Always select appropriate antibodies for your research and where possible validate them yourself. After checking vendor details about the intended applications and whether they match your planned applications, also think about whether you will need a long-term supply of the antibody. Polyclonal antibodies can have variability between batches and while monoclonal antibodies suffer from this to a far lesser extent, genetic drift in hybridomas can also result in batch variability over time. Recombinant technology can help overcome this limitation by providing antibodies in a range of formats with an unlimited supply. For example, Bio-Rad’s recombinant monoclonal antibodies for cancer research are well-characterized and have consistent lot-to-lot performance making them an ideal choice for longitudinal studies.
When you publish your research, include all relevant information about your antibody choice, including any information about how you validated the antibody for the chosen applications to enable other researchers to have confidence in your data.
How Is Bio-Rad Tackling the Reproducibility Crisis?
We are committed to evolving our validation standards to ensure quality and reproducibility, and develop rigorous validation guidelines benefiting the scientific community as a whole. We conduct thorough in-house testing and clearly display recommended applications for every antibody, and as more of our antibodies are evaluated in our ongoing enhanced validation program, those that meet our stringent validation criteria earn the PrecisionAb Antibody label.
References
Baker M (2015). Reproducibility crisis: blame it on the antibodies. Nature 521, 274–276.
Baker M (2020). When antibodies mislead: the quest for validation. Nature 585, 313–314.
Fonseca SG et al. (2015). Retraction Note: Wolfram syndrome 1 and adenylyl cyclase 8 interact at the plasma membrane to regulate insulin production and secretion. Nat Cell Bio 17, 105.
Lyst MJ et al. (2015). Retraction: Regulation of MBD1‐mediated transcriptional repression by SUMO and PIAS proteins. EMBO J 38, e103220.
Vasilevsky NA et al. (2013). On the reproducibility of science: unique identification of research resources in the biomedical literature. PeerJ 1, e148.
Uhlen M et al. (2016). A proposal for validation of antibodies. Nat Methods 13, 823–827.
Weller MG (2018). Ten basic rules of antibody validation. Anal Chem Insights, 13, 1177390118757462.
You may also be interested in...

View more applications blogs














