5 Easy Steps for Successful Flow Cytometry
- Mar 08, 2016
- 8 min read
- Bio-Rad
Flow Cytometry is a powerful tool for analyzing the physical and chemical properties of cells and other components such as nuclei and chromosomes. However, if you are a Flow Cytometry beginner, the whole process can be quite confusing. In order to get the best results, a lot of forethought and planning is required. In this blog post we simplify the process in five easy steps that will ensure successful results.
Step 1:
Plan your experiment in advance
As is the case with most experiments, careful planning beforehand goes a long way in generating reproducible results. For successful Flow Cytometry however, this is even more important. To guide you in your planning, here are a few questions you should carefully consider before beginning your experiment:
Are there reagents available for your target proteins or cells of interest?
We recommend that you first do a literature search or use the antibody search engine Citeab, which collates the most commonly cited antibodies for your specific targets. On Citeab you can also search by application, to ensure that you use the right clone for a particular application.
Is your flow cytometer suitable for the fluorophores you are using?
It is important that you first check the configuration of your cytometer to determine whether there are available lasers for the fluorophores you would like to use. In addition, check the excitation and emission spectra of each fluorophore to assess spillover, which is fluorescence from one fluorophore spilling into the detection channel of another fluorophore (see more tips on panel design). Use a fluorescence spectra viewer to assist with this, and try to choose fluorophores with as little spillover as possible. Knowing the relative brightness of prospective fluorophores will also help you select the best panel. You can find a fluorophore brightness chart and more information in our choosing the right fluorophores article.
What is the cell biology and expected experimental outcome?
Understanding the biology of your experimental samples is critical for successful Flow Cytometry results. For instance, knowing the relative expression of your target antigen will help you determine which fluorophore to use for which marker. The rule of thumb is to use dim fluorophores with highly expressed antigens and brighter fluorophores for antigens with lower expression. If you are unsure of the expression of a target antigen under your experimental conditions, use a fluorophore that has little spill over into other channels. In addition, knowing the population density or frequency of your target cells is critical for determining the number of events to collect or starting sample to generate in order to obtain meaningful results (see table below). Other things to be aware of are cellular changes, interfering components, any changes in marker expression level as well as whether there are any cellular interactions involving the cell population you are analyzing.
Starting population |
Frequency |
Number of cells analyzed |
|---|---|---|
|
1,000,000 |
10% |
100,000 |
|
1,000,000 |
1% |
10,000 |
|
1,000,000 |
0.1% |
1,000 |
Step 2:
Carefully prepare your cells
Now that you have done all the planning, the next step involves careful preparation of your samples. So, what is your starting sample?
Depending on your research question several sample types are often used in Flow Cytometry analysis, and each requires unique handling. For each sample type the key is to retain cell viability and generate a single cell suspension. Here are a few tips to consider for the most common sample types used:
Cell culture
Determine whether the cells are adherent or suspension cells, as this will help you decide the most careful method of harvesting them for analysis. In deciding which method is right for you, the goal is to choose the least harsh treatment that gives the best viability and does not affect cellular function. For example, trypsin-EDTA or Accutase™ can be used to harvest adherent cells, however if your target antigen is expressed on the cell surface, trypsin may not be ideal as this enzyme cleaves cell surface antigens. Cell scraping can also be used to harvest adherent cells but this method may result in decreased cell viability. In preparing your cells, Ca2+ or Mg2+ free medium should be used to avoid cell clumping since these elements are required by cell surface integrins to bind proteins and substrates, thus increasing clumping.
Bone marrow cells
When collecting bones, try to remove as much muscle and tissue as possible as these can clog the flow cytometer and also bind non-specifically to your antibody. In addition, try to avoid contaminants by flushing bones under sterile conditions.
Secondary lymphoid organs (spleen, thymus or lymph nodes)
Homogenization can be used to harvest cells from these organs, however it is important not to over homogenize as this can lead to DNA release from cells, which can cause clumping. Adding EDTA or DNase I to the media can help to control the clumping from homogenization. For these tissues however, more gentle techniques such as mashing through a cell strainer with a syringe plunger may be necessary.
Solid organs
Homogenization with the addition of enzymatic digestion is often used for solid organs. This breaks down the extracellular matrix, thus releasing the cells. Therefore, the enzyme used is tissue specific, and without it, the cells might not release from the organ. Being careful to avoid the release of sticky DNA and cell clumping is also important for harvesting cells from solid organs. Again, addition of DNase I or EDTA will assist with generating a single cell suspension.
Blood
Using an appropriate anti-coagulant is critical when preparing blood for Flow Cytometry analysis. For example, EDTA ruins antigen conformation and is not suitable for intracellular staining of cytokines. It should also be noted that if you use Ficoll™ to separate blood components you will miss the granulocytes if you are interested in studying this cell population.
Step 3:
Control, control, and control
Setting up your experiments with proper controls is necessary for making sense of your data in the end. It will help you to determine what is truly negative or positive, and how to set up your gates for analysis. Plan to include the following controls in your experimental design:
Unstained
These are cells that do not have any antibody staining and provides an indication of negative staining and background level of fluorescence.
Single stained
These are necessary for setting up your compensation controls.
Unstimulated/Fully stimulated
This is particularly important for cytokine release assays because it allows you to determine the dynamic range of your experiment.
Known negative/positive
If you are studying a cell population for the first time, this set of controls will give you an estimation of what to expect and where cells should be positioned in your gates.
Isotype controls
The use of isotype controls is quite controversial (as discussed in our multi-color flow panel control article); however they can assist in providing an estimation of non-specific binding through the Fc receptor as well as non-specific surface binding of the antibody and the fluorophore.
Fluorescence minus one (FMO)
These controls are popular when building multicolor panels as they provide information on fluorophore spread and cross laser stimulation.
Step 4:
Make it count- Get the most out of your staining
To ensure you get the best staining, several factors need to be considered. Here are the most important things to do before your antibody staining steps to improve your results:
Blocking non-specific binding
Since non-specific binding can significantly impact data analysis and conclusions, adding serum such as BSA or FCS to your buffer as well as including an Fc blocking step prior to antibody staining will improve your results.
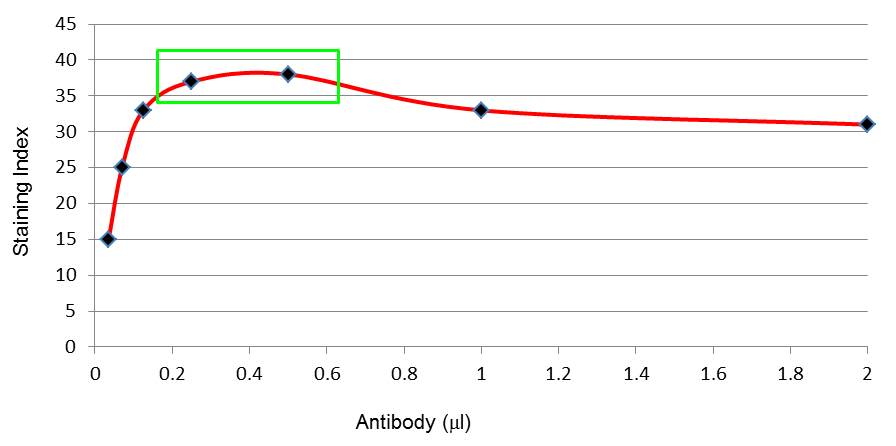
Antibody titration
Excess antibodies will bind at low affinity and create background that will cloud your results. Therefore, it is important to determine the right amount of antibody needed for your specific sample. If you use isotype controls, be sure to use them at the same concentration as your proper antibody. To determine the best antibody concentration, the stain index, which is defined as the ratio of the separation between the positive and negative population divided by two times the standard deviation of the negative population, can be used as a guide. Calculating the stain index for each antibody concentration tested and generating a graph similar to the one below (antibody concentration vs staining index) will help you decide on the right antibody concentration. In the graph below, the points in the green box represent the ideal concentrations that will generate specific staining with the least amount of background.
Stain Index:
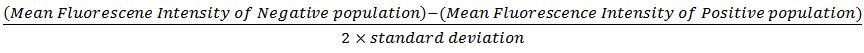
Step 5:
Remove doublets and dead cells
Doublets can significantly affect the accuracy of your results by including false positives into your analysis. As cells pass through the laser beam, a doublet of a negative cell and a positive cell will produce the same fluorescence as a single positive cell leading to the inadvertent inclusion of a negative cell into your analysis (see image below).
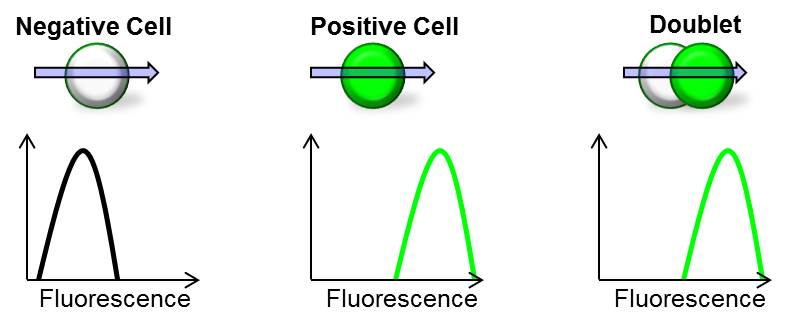
For immunophenotyping experiments, if a FITC positive cell stuck to a PE positive cell, the flow cytometer would register one cell that was double positive, which would be inaccurate. Furthermore, in cell cycle analysis, doublets will misrepresent 4n cells, leading you to make incorrect conclusions.
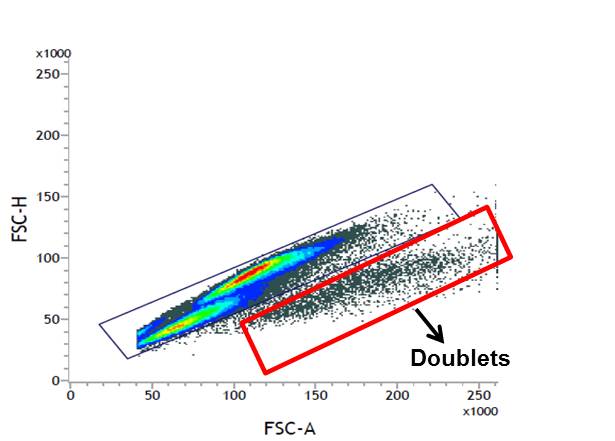
Therefore, to ensure the accuracy of your results, doublets should be excluded during analysis. One way to do this is by using the forward scatter height (FSC-H) vs forward scatter area (FSC-A) method (see image below). The height measurement denotes the intensity of the signal (i.e. expression of your target protein) and the area correlates signal/protein expression with cell size. Therefore, cells of the same size will generally show very similar area and height on a dot plot. As shown in the dot plot below, singlets are separated from doublets and clumps and are clustered diagonally. Doublets can therefore by excluded because they will have an abnormally larger area. Be sure to set the area scaling feature on your flow cytometer, using your cells not beads, prior to analysis in order to correctly exclude doublets using this method.
Dead cells can also seriously affect your analysis. These cells are autofluorescent and can lead to increased background through non-specific binding as well as reduce the dynamic range so that weak positive samples will not be identifiable. Therefore it is highly recommended that you always include a dead cell marker in your experimental design. It is not sufficient to just exclude the cell debris at the bottom of the FSC/SSC gate from your analysis as dead cells could be included with your gated samples.
Further your flow skills
There you have it. These steps serve as a basic guideline for successful Flow Cytometry results. To further improve your Flow Cytometry skills, we have an array of resources to help you become a Flow master, including protocols, troubleshooting tips, intracellular flow cytometry tips and a handy Introduction to Flow Cytometry guide.
Also check out our Flow Cytometry webinar, which provides further details on optimizing your flow cytometry.
Ficoll is a registered trademark owned by GE Healthcare companies.