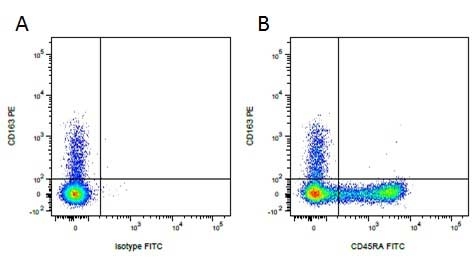
CD45RA antibody | MIL13





Mouse anti Pig CD45RA:RPE
- Product Type
- Monoclonal Antibody
- Clone
- MIL13
- Isotype
- IgG1
- Specificity
- CD45RA
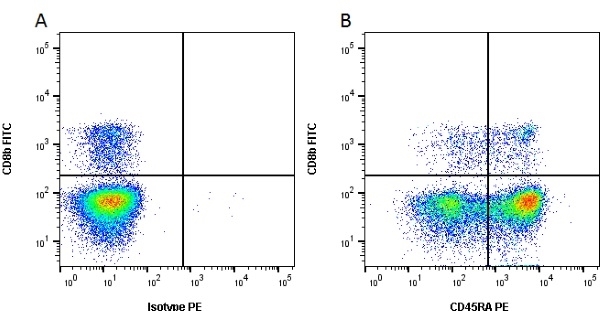
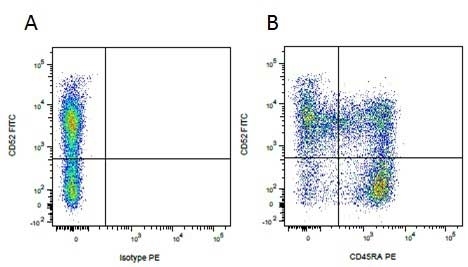
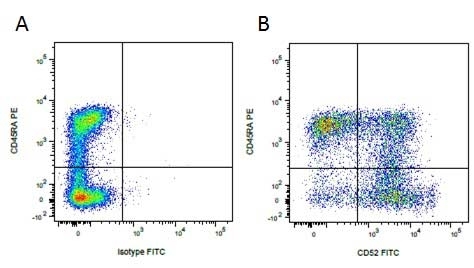
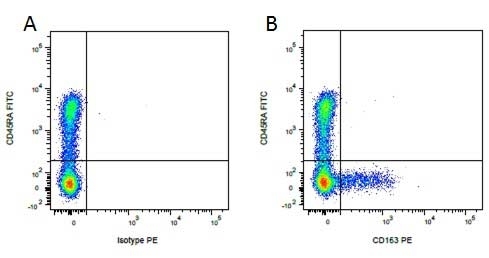
| Mouse anti Pig CD45RA, clone MIL13, recognizes an epitope contained in the portion of porcine CD45 encoded by exon A, CD45RA (Lunney et al. 2007). Mouse anti pig CD45RA, clone MIL13 recognizes both the 210 kDa RA CD45 isoform and the 226 kDa RAC isoform (Zuckermann et al. 2001). Clone MIL13 does not recognize the CD45RC or CD45RO isoforms. |
- Target Species
- Pig
- Product Form
- Purified IgG conjugated to R. Phycoerythrin (RPE) - lyophilized
- Reconstitution
- Reconstitute with 1.0 ml distilled water
- Preparation
- Purified IgG prepared from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
1% bovine serum albumin
5% sucrose - Immunogen
- Cells isolated from porcine mesenteric lymph node
- Max Ex/Em
-
Fluorophore Excitation Max (nm) Emission Max (nm) RPE 488nm laser 496 578 - Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
DO NOT FREEZE.
This product should be stored undiluted. This product is photosensitive and should be protected from light. Should this product contain a precipitate we recommend microcentrifugation before use.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | Neat |
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 106 cells in 100μl
How to Use the Spectraviewer
Watch the Tool Tutorial Video ▸- Start by selecting the application you are interested in, with the option to select an instrument from the drop down menu or create a customized instrument
- Select the fluorophores or fluorescent proteins you want to include in your panel to check compatibility
- Select the lasers and filters you wish to include
- Select combined or multi-laser view to visualize the spectra
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control:RPE | MCA928PE | F | 100 Tests | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | Mouse IgG1 Negative Control:RPE | ||||||
References for CD45RA antibody
-
Pakkanen, T.M. et al. (2000) Periadventitial lacZ gene transfer to pig carotid arteries using a biodegradable collagen collar or a wrap of collagen sheet with adenoviruses and plasmid-liposome complexes.
J Gene Med. 2: 52-60. -
Terzic, S. et al. (2002) Immunophenotyping of leukocyte subsets in peripheral blood and palatine tonsils of prefattening pigs.
Vet Res Commun. 26: 273-83. -
Bozić F et al. (2002) Recruitment of intestinal CD45RA+ and CD45RC+ cells induced by a candidate oral vaccine against porcine post-weaning colibacillosis.
Vet Immunol Immunopathol. 86 (3-4): 137-46. -
Schierack, P. et al. (2009) Effects of Bacillus cereus var. toyoi on immune parameters of pregnant sows.
Vet Immunol Immunopathol. 127: 26-37. -
Thierry, A. et al. (2012) Identification of invariant natural killer T cells in porcine peripheral blood.
Vet Immunol Immunopathol. 149 (3-4): 272-9. -
Suzuki, S. et al. (2016) Generation and characterization of RAG2 knockout pigs as animal model for severe combined immunodeficiency.
Vet Immunol Immunopathol. 178: 37-49. -
López, E. et al. (2019) Identification of very early inflammatory markers in a porcine myocardial infarction model.
BMC Vet Res. 15 (1): 91. -
Li, K.et al. (2019) Generation of porcine monoclonal antibodies based on single cell technologies.
Vet Immunol Immunopathol. 215: 109913.
View The Latest Product References
-
Forner, R. et al. (2021) Distribution difference of colostrum-derived B and T cells subsets in gilts and sows.
PLoS One. 16 (5): e0249366. -
Ogihara, K. et al. (2022) A porcine lymphoma-derived cell line co-expressing IgM, IgG and IgA.
J Vet Med Sci. 84 (6): 760-5. -
Zhao, H. et al. (2022) Development of RAG2 -/- IL2Rγ -/Y immune deficient FAH-knockout miniature pig.
Front Immunol. 13: 950194. -
Haach, V. et al. (2023) A polyvalent virosomal influenza vaccine induces broad cellular and humoral immunity in pigs.
Virol J. 20 (1): 181. -
Li, J. et al. (2024) Single-cell transcriptomic analysis reveals transcriptional and cell subpopulation differences between human and pig immune cells.
Genes Genomics. 46 (3): 303-22.
Further Reading
-
Piriou-Guzylack, L. (2008) Membrane markers of the immune cells in swine: an update.
Vet Res. 39: 54.
- RRID
- AB_323626
MCA1751PE
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Pig ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up