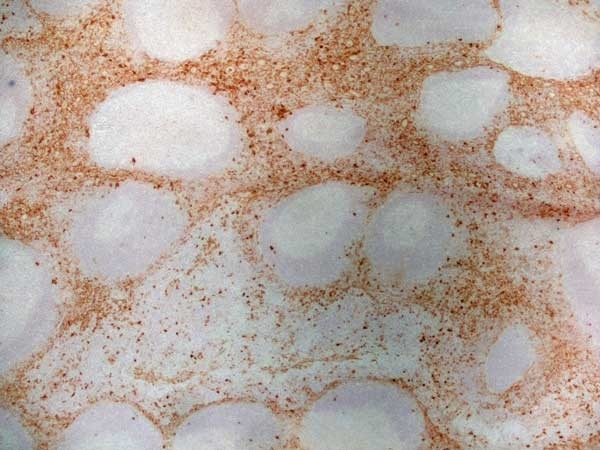
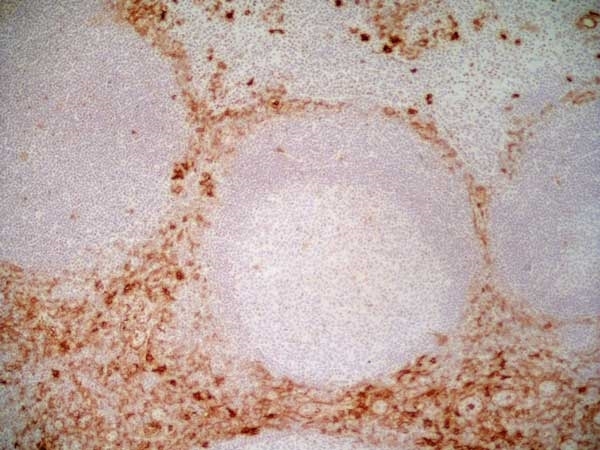
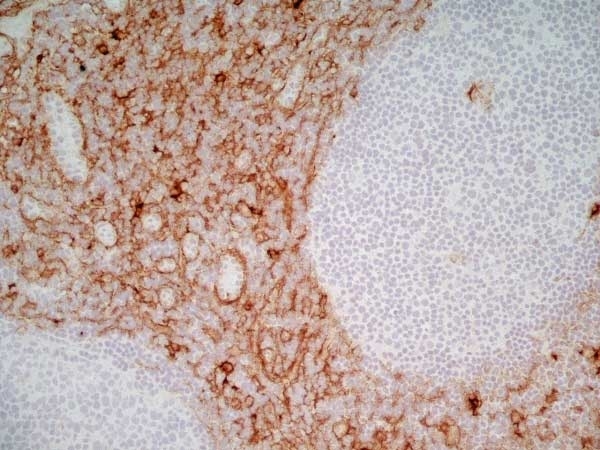
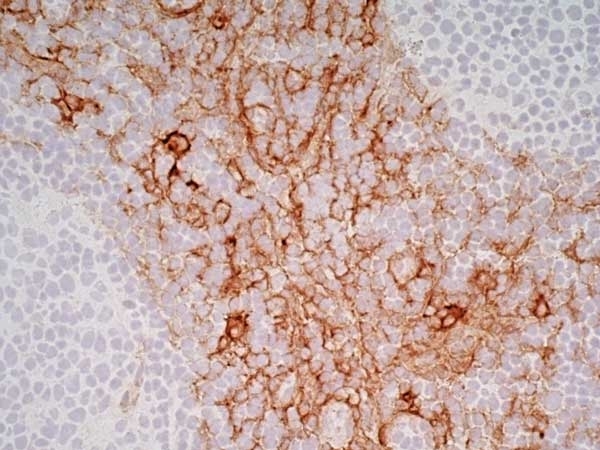
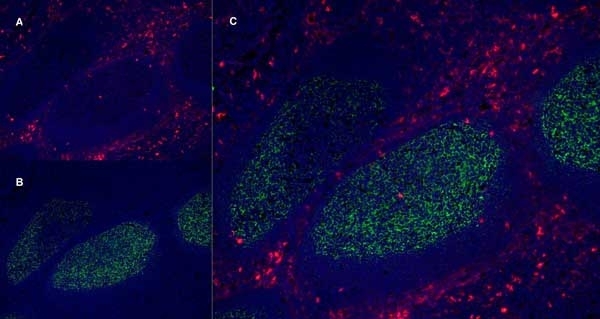
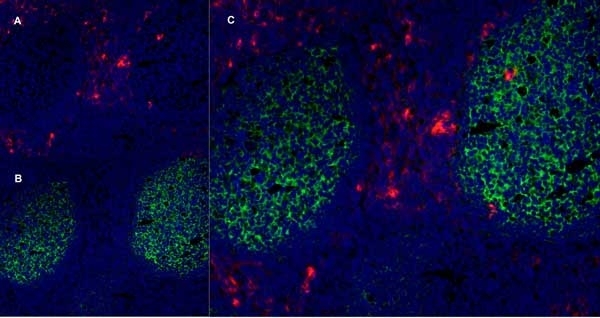
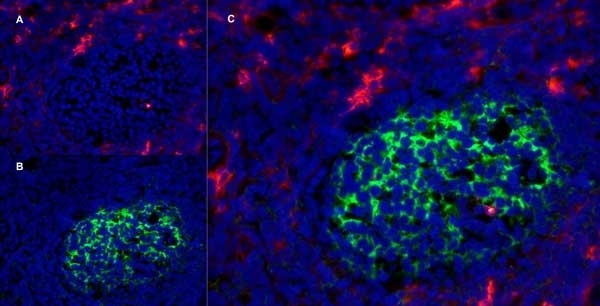
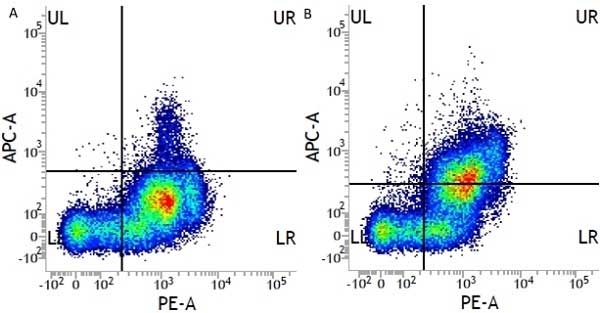
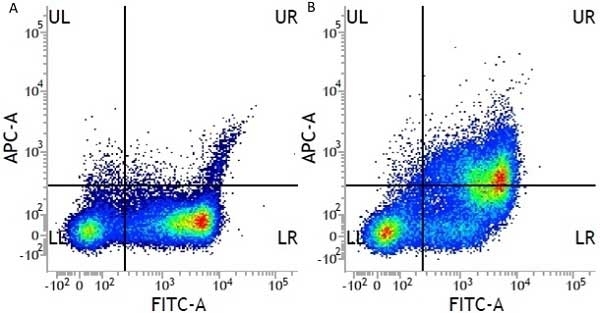
CD13 antibody | WM15


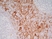






Mouse anti Human CD13
- Product Type
- Monoclonal Antibody
- Clone
- WM15
- Isotype
- IgG1
- Specificity
- CD13
| Mouse anti Human CD13 antibody, clone WM15 recognizes human CD13 also known as aminopeptidase N. CD13 is a single pass type II glycosylated integral membrane protein with a predicted molecular mass of ~110 kDa and an apparent molecular mass of ~150 kDa expressed by granulocytes, monocytes, fibroblasts, endothelial cells and by myeloid leukaemia cells (Bradstock et al. 1985). CD13 acts as a major cell surface receptor for group 1 coronoviruses (Breslin et al. 2003) which bind to a critical sequence encompassing amino acid residies 288-295 (Kolb et al. 1997). CD13 functions as an aminopeptidase enzyme, a metalloprotease present as both a membrane bound form and also a soluble aminopeptidase N. Mouse anti Human CD13, clone WM15 inhibits infection of cells by human coronavirus (Lachance et al. 1998) but not hepatitis C virus (Koutsoudakis et al. 2006) and inhibits aminopeptidase N activity of the CD13 molecule (Asmun et al. 1992). |
- Target Species
- Human
- Species Cross-Reactivity
-
Target Species Cross Reactivity Rhesus Monkey - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
- Carrier Free
- Yes
- Immunogen
- Human AML cells.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunized BALB/c mice where fused with cells of the mouse NS1 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| ELISA | |||
| Flow Cytometry | 1/50 | 1/100 | |
| Immunohistology - Frozen | |||
| Immunoprecipitation | |||
| Western Blotting |
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 106 cells or 100μl whole blood
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA928 | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
Source Reference
-
Bradstock, K.F. et al. (1985) Human myeloid differentiation antigens identified by monoclonal antibodies: expression on leukemic cells.
Pathology. 17 (3): 392-9.
References for CD13 antibody
-
Bradstock, K.F. et al. (1985) Myeloid progenitor surface antigen identified by monoclonal antibody.
Br J Haematol. 61 (1): 11-20. -
Favaloro, E.J. et al. (1988) Further characterization of human myeloid antigens (gp160,95; gp150; gp67): investigation of epitopic heterogeneity and non-haemopoietic distribution using panels of monoclonal antibodies belonging to CD-11b, CD-13 and CD-33.
Br J Haematol. 69 (2): 163-71. -
Favaloro, E.J. (1991) CD-13 (gp150; aminopeptidase-N): co-expression on endothelial and haemopoietic cells with conservation of functional activity.
Immunol Cell Biol. 69 ( Pt 4): 253-60. -
Favaloro, E.J. et al. (1993) The hepatobiliary disease marker serum alanine aminopeptidase predominantly comprises an isoform of the haematological myeloid differentiation antigen and leukaemia marker CD-13/gp150.
Clin Chim Acta. 220 (1): 81-90. -
Favaloro, E.J. et al. (1993) CD13 (GP150; aminopeptidase-N): predominant functional activity in blood is localized to plasma and is not cell-surface associated.
Exp Hematol. 21 (13): 1695-701. -
Gredmark, S. et al. (2004) Human Cytomegalovirus Induces Inhibition of Macrophage Differentiation by Binding to Human Aminopeptidase N/CD13
J Immunol. 173: 4897-907 -
Thielitz, A. et al. (2004) Identification of extra- and intracellular alanyl aminopeptidases as new targets to modulate keratinocyte growth and differentiation.
Biochem Biophys Res Commun. 321 (4): 795-801. -
Lassnig, C. et al. (2005) Development of a transgenic mouse model susceptible to human coronavirus 229E.
Proc Natl Acad Sci U S A. 102 (23): 8275-80.
View The Latest Product References
-
Stolzing, A. et al. (2008) Age-related changes in human bone marrow-derived mesenchymal stem cells: consequences for cell therapies.
Mech Ageing Dev. 129: 163-73. -
Negussie, A.H. et al. (2010) Synthesis and in vitro evaluation of cyclic NGR peptide targeted thermally sensitive liposome.
J Control Release. 143: 265-73. -
Grzywacz, B. et al. (2011) Natural killer-cell differentiation by myeloid progenitors.
Blood. 117: 3548-58. -
Tavoosidana, G. et al. (2011) Multiple recognition assay reveals prostasomes as promising plasma biomarkers for prostate cancer.
Proc Natl Acad Sci U S A. 108: 8809-14. -
Silk, K.M. et al. (2012) Rapamycin conditioning of dendritic cells differentiated from human ES cells promotes a tolerogenic phenotype.
J Biomed Biotechnol. 2012:172420. -
McCormack, E. et al. (2013) Multiplexed mAbs: a new strategy in preclinical time-domain imaging of acute myeloid leukemia.
Blood. 121 (7): e34-42. -
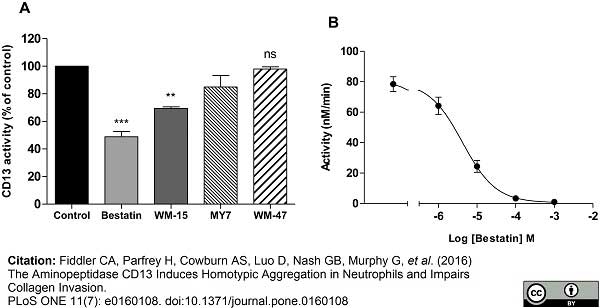
Fiddler, C.A. et al. (2016) The Aminopeptidase CD13 Induces Homotypic Aggregation in Neutrophils and Impairs Collagen Invasion.
PLoS One. 11 (7): e0160108. -
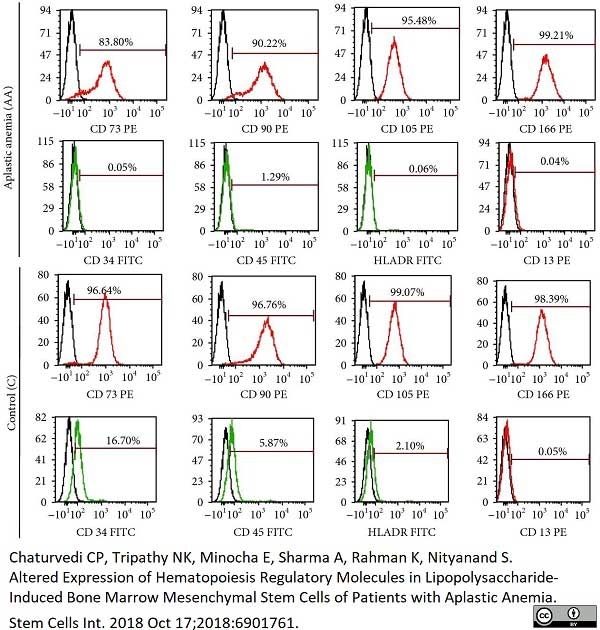
Chaturvedi, C.P. et al. (2018) Altered Expression of Hematopoiesis Regulatory Molecules in Lipopolysaccharide-Induced Bone Marrow Mesenchymal Stem Cells of Patients with Aplastic Anemia.
Stem Cells Int. 2018: 6901761. -
Menon, R. et al. (2023) Human Induced Pluripotent Stem Cell-Derived Pericytes as Scalable and Editable Source to Study Direct Lineage Reprogramming Into Induced Neurons.
Cell Reprogram. 25 (5): 212-23. -
Karpyuk, V. et al. (2019) Innovation-based Approach in Reconstruction of Reduced Jaw Alveolar Ridge Bone Using Cell Regeneration Technologies
Archiv Euromedica 9 (2) 147-55.
- Synonyms
- Aminopeptidase N
- RRID
- AB_321309
- UniProt
- P15144
- Entrez Gene
- ANPEP
- GO Terms
- GO:0001525 angiogenesis
- GO:0004177 aminopeptidase activity
- GO:0008237 metallopeptidase activity
- GO:0005829 cytosol
- GO:0004872 receptor activity
- GO:0005887 integral to plasma membrane
- GO:0005793 ER-Golgi intermediate compartment
- GO:0008270 zinc ion binding
- GO:0030154 cell differentiation
- View More GO Terms
- GO:0044419 interspecies interaction between organisms
MCA1270
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Human ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up