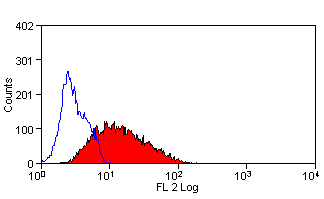
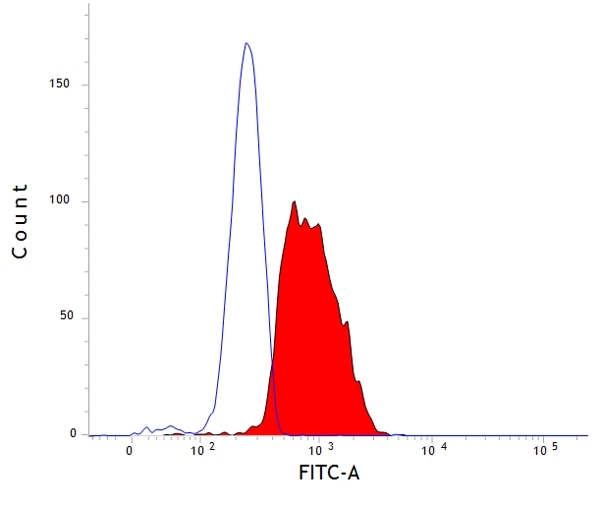
CD107b antibody | AC17


Mouse anti Dog CD107b
- Product Type
- Monoclonal Antibody
- Clone
- AC17
- Isotype
- IgG1
- Specificity
- CD107b
| Mouse anti Dog CD107b antibody, clone AC17 recognizes canine CD107b, otherwise known as lysosome-associated membrane protein 2 or LAMP-2. Immunofluorescence staining of MDCK cells with mouse anti dog CD107b, clone AC17 demonstrates staining patterns consistent with localization to lysozomes. This is supported by coincident staining of an exogenous lysozomal glycoprotein, avian LEP100 transfected into MDCK cells and detected using the anti LEP100 antibody clone CV24 (Nabi et al.1991). Mouse anti Dog CD107b antibody, clone AC17 immunoprecipitates a protein of ~95 kDa in MDCK cells which, following Endo F digestion to remove N-linked oligosaccharides, yields a core protein product of 40 kDa, indicating the heavily glycosylated nature of CD107b. The molecular weight of canine CD107b is typical of many lysozome-associated membrane proteins. While most (97%) CD107b resides in the lysozomal environment in adherent MDCK cells in vitro, a small percentage is associated with the cell membrane (Nabi et al.1991). CD107b has been shown to share high N-terminal amino acid sequence homology with human, mouse and rat CD107b (Nabi et al.1993). Transfection of a mink type II lung epithelial cell line with beta1-6-N-acetylglucosaminyl transferase V demonstrates the formation of large lysozomal vacuoles, termed multilamellar bodies (MLBs), having a very distinct phenotype with expression of CD107b, as indicated by immunofluorescent staining with clone AC17. These MLBs require lysozomal degradation via an autophagic pathway for their formation and may have implications for lysozomal storage diseases (Hariri et al.2000). CD107b is involved in the lysosomal uptake of cytosolic proteins and the endocytic pathway. Mouse anti Dog CD107b antibody, clone AC17 is suitable for use in electron microscopy (Nabi et al.1991). |
- Target Species
- Dog
- Species Cross-Reactivity
-
Target Species Cross Reactivity Mouse Mink Human Rat - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
0.09% Sodium Azide (NaN3) - Carrier Free
- Yes
- Immunogen
- MDCK (Madin-Darby Canine Kidney) cells
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunised Balb/c mice were fused with cells of the NS1 myeloma cell line
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry 1 | 1/50 | 1/200 | |
| Immunofluorescence | |||
| Immunoprecipitation | |||
| Western Blotting | 1/100 | 1/500 |
- 1 Membrane permeabilization is required for this application. The use of Leucoperm (Product Code BUF09) is recommended for this purpose.
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 1x106 cells in 100ul
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA928 | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
References for CD107b antibody
-
Nabi, I.R. et al. (1991) An endogenous MDCK lysosomal membrane glycoprotein is targeted basolaterally before delivery to lysosomes.
J Cell Biol. 115 (6): 1573-84. -
Nabi, I.R. & Rodriguez-Boulan, E. (1993) Increased LAMP-2 polylactosamine glycosylation is associated with its slower Golgi transit during establishment of a polarized MDCK epithelial monolayer.
Mol Biol Cell. 4 (6): 627-35. -
Hariri, M. et al. (2000) Biogenesis of multilamellar bodies via autophagy.
Mol Biol Cell. 11: 255-68. -
Jou, T.S. et al. (2000) Selective alterations in biosynthetic and endocytic protein traffic in Madin-Darby canine kidney epithelial cells expressing mutants of the small GTPase Rac1.
Mol Biol Cell. 11 (1): 287-304. -
Ihrke, G. et al. (2001) Competing sorting signals guide endolyn along a novel route to lysosomes in MDCK cells.
EMBO J. 20 (22): 6256-64. -
Cliffe, S.T. et al. (2009) SLC29A3 gene is mutated in pigmented hypertrichosis with insulin-dependent diabetes mellitus syndrome and interacts with the insulin signaling pathway.
Hum Mol Genet. 18: 2257-65. -
Pluhar, G.E. et al. (2010) Anti-tumor immune response correlates with neurological symptoms in a dog with spontaneous astrocytoma treated by gene and vaccine therapy.
Vaccine 28 (19): 3371-8. -
Nagahama, M. et al. (2011) Cellular vacuolation induced by Clostridium perfringens epsilon-toxin.
FEBS J. 278: 3395-407.
View The Latest Product References
-
Bai, Y. et al. (2011) Intracellular neutralization of viral infection in polarized epithelial cells by neonatal Fc receptor (FcRn)-mediated IgG transport.
Proc Natl Acad Sci U S A. 108 (45): 18406-11. -
Nagahama, M. et al. (2012) Intracellular trafficking of Clostridium perfringens iota-toxin b.
Infect Immun. 80: 3410-6.
Further Reading
-
Fukuda, M. (1991) Lysosomal membrane glycoproteins. Structure, biosynthesis, and intracellular trafficking.
J Biol Chem. 266 (32): 21327-30.
- Synonyms
- LAMP-2
MCA2558GA
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Dog ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up