CD14 antibody | CC-G33




Mouse anti Bovine CD14:FITC
- Product Type
- Monoclonal Antibody
- Clone
- CC-G33
- Isotype
- IgG1
- Specificity
- CD14
| Mouse anti Bovine CD14, clone CC-G33 recognizes bovine CD14. CD14 is a GPI-anchored membrane glycoprotein and monocyte/macrophage differentiation antigen, belonging to the lipopolysaccharide receptor family, also expressed weakly on microglia and Langerhans cells. CD14 acts as a receptor for the potent bacterial endotoxin, lipopolysaccharide (LPS), facilitated by LPS-binding protein (LBP). The binding of LPS to CD14 results in cell activation and the release of cytokines and the inflammatory response, and has been shown to upregulate the cell surface expression of adhesion molecules. Mouse anti Bovine CD14 clone CC-G33 cross-reacts with human CD14 expressed on transfected COS-7 cells (Berthon & Hopkins 1996), ovine CD14 (Sopp et al. 1996) and Water buffalo (Bubalus bubalis) CD14, (Mirielli et al. 2013). |
- Target Species
- Bovine
- Species Cross-Reactivity
-
Target Species Cross Reactivity Sheep Human Water Buffalo - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG conjugated to Fluorescein Isothiocyanate Isomer 1 (FITC) - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
0.09% Sodium Azide (NaN3) 1% Bovine Serum Albumin - Immunogen
- Partially purified polypeptides isolated from bovine leucocyte cell surface membrane.
- Approx. Protein Concentrations
- IgG concentration 0.1mg/ml
- Fusion Partners
- Spleen cells from immunized Balb/c mice were fused with cells of the NS1 myeloma cell line.
- Max Ex/Em
-
Fluorophore Excitation Max (nm) Emission Max (nm) FITC 490 525 - Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended. This product is photosensitive and should be protected from light.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry |
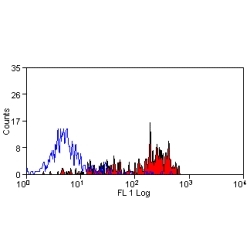
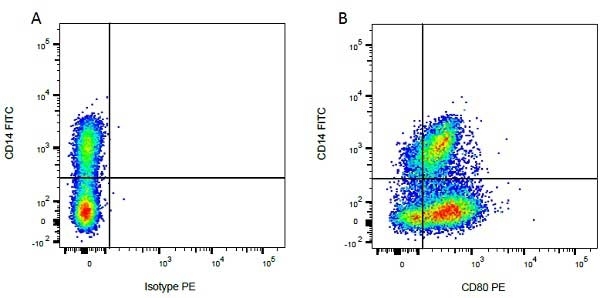
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 1x106 cells in 100ul.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control:FITC | MCA928F | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control:FITC | ||||||
Source Reference
-
Sopp, P. et al. (1996) Identification of bovine CD14.
Vet Immunol Immunopathol. 52 (4): 323-8.
References for CD14 antibody
-
Berthon, P. & Hopkins, J. (1996) Ruminant cluster CD14.
Vet Immunol Immunopathol. 52 (4): 245-8. -
Haas, K.M. and Estes, D.M. (2001) The identification and characterization of a ligand for bovine CD5.
J Immunol. 166: 3158-66. -
Altreuther, G. et al. (2001) Morphologic and functional changes in bovine monocytes infected in vitro with the bovine leukaemia virus.
Scand J Immunol. 54: 459-69. -
Fikri Y et al. (2002) Costimulatory molecule requirement for bovine WC1+gammadelta T cells' proliferative response to bacterial superantigens.
Scand J Immunol. 55 (4): 373-81. -
Glew, E.J. et al. (2003) Differential effects of bovine viral diarrhoea virus on monocytes and dendritic cells.
J Gen Virol. 84: 1771-80. -
Harris, J. et al. (2003) Expression of caveolin by bovine lymphocytes and antigen-presenting cells.
Immunology. 105: 190-5. -
Villarreal-Ramos, B. et al. (2003) Investigation of the role of CD8+ T cells in bovine tuberculosis in vivo.
Infect Immun.71: 4297-303. -
Gliddon, D.R. et al. (2004) DEC-205 expression on migrating dendritic cells in afferent lymph.
Immunology. 11: 262-72.
View The Latest Product References
-
Villarreal-Ramos, B. et al. (2006) Influence of the nature of the antigen on the boosting of responses to mycobacteria in M. bovis-BCG vaccinated cattle.
Vaccine. 24 (47-48): 6850-8. -
Herath, S. et al. (2006) Expression and function of Toll-like receptor 4 in the endometrial cells of the uterus.
Endocrinology. 147: 562-70. -
Yamakawa, Y. et al. (2008) Identification and functional characterization of a bovine orthologue to DC-SIGN.
J Leukoc Biol. 83: 1396-403. -
Pirson, C. et al. (2012) Differential effects of Mycobacterium bovis - derived polar and apolar lipid fractions on bovine innate immune cells.
Vet Res. 43: 54. -
Gibson, A. et al. (2012) Identification of a lineage negative cell population in bovine peripheral blood with the ability to mount a strong type I interferon response
Dev Comp Immunol. 36: 332-41. -
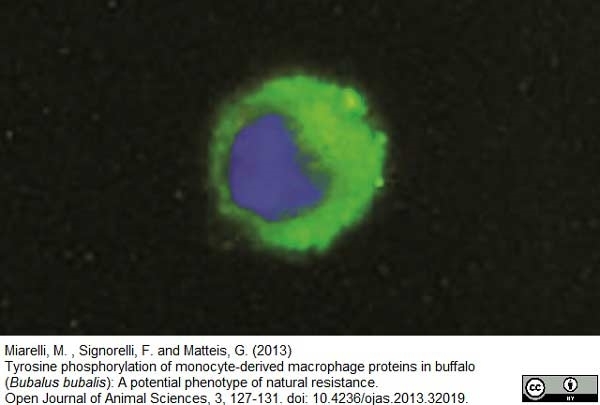
Miarelli, M. et al. (2013) Tyrosine phosphorylation of monocyte-derived macrophage proteins in buffalo (Bubalus bubalis): A potential phenotype of natural resistance
Open J Anim Sci. 03 (02): 127-31. -
Hecker YP et al. (2014) A Neospora caninum vaccine using recombinant proteins fails to prevent foetal infection in pregnant cattle after experimental intravenous challenge.
Vet Immunol Immunopathol. 162 (3-4): 142-53. -
Brodzki, P. et al. (2014) Phenotyping of leukocytes and granulocyte and monocyte phagocytic activity in the peripheral blood and uterus of cows with endometritis.
Theriogenology. 82 (3): 403-10. -
Vrieling, M. et al. (2015) Bovine Staphylococcus aureus Secretes the Leukocidin LukMF' To Kill Migrating Neutrophils through CCR1.
MBio. 6 (3): e00335. -
Herry, V. et al. (2017) Local immunization impacts the response of dairy cows to Escherichia coli mastitis.
Sci Rep. 7 (1): 3441. -
Pepponi, I. et al. (2017) A mycobacterial growth inhibition assay (MGIA) for bovine TB vaccine development.
Tuberculosis (Edinb). 106: 118-22. -
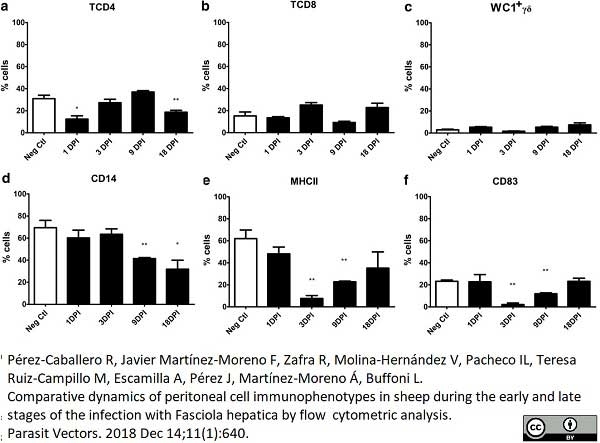
Pérez-caballero, R. et al. (2018) Comparative dynamics of peritoneal cell immunophenotypes in sheep during the early and late stages of the infection with Fasciola hepatica by flow cytometric analysis.
Parasit Vectors. 11 (1): 640. -
de Araújo, F.F.et al. (2019) Distinct immune response profile during Rhipicephalus (Boophilus) microplus. infestations of guzerat dairy herd according to the maternal lineage ancestry (mitochondrial DNA).
Vet Parasitol. 273: 36-44. -
Oliveira, B.M. et al. (2020) Characterization of Myeloid Cellular Populations in Mesenteric and Subcutaneous Adipose Tissue of Holstein-Friesian Cows.
Sci Rep. 10 (1): 1771. -
Liu, J. et al. (2020) Theileria annulata. transformation altered cell surface molecules expression and endocytic function of monocyte-derived dendritic cells.
Ticks Tick Borne Dis. 11 (3): 101365. -
Wu, Y. et al. (2019) Changes in the small intestine mucosal immune barrier in Muscovy ducklings infected with Muscovy duck reovirus.
Vet Microbiol. 233: 85-92. -
Broberg, L. et al. (2021) Isolation and characterization of eosinophils in bovine blood and small intestine.
Vet Immunol Immunopathol. 242: 110352. -
Andrés, S. et al. (2024) Essential oil supplementation in milk replacers: short- and long-term impacts on feed efficiency, the faecal microbiota and the plasma metabolome in dairy calves.
J Dev Orig Health Dis. : 1-11.
- RRID
- AB_1604717
- UniProt
- Q95122
- Entrez Gene
- CD14
- GO Terms
- GO:0005886 plasma membrane
- GO:0006954 inflammatory response
- GO:0031225 anchored to membrane
- GO:0045087 innate immune response
MCA2678F
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Bovine ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up