Caspase-1

FAM FLICA™ Caspase-1 Kit
- Product Type
- Kits
- Specificity
- Caspase-1
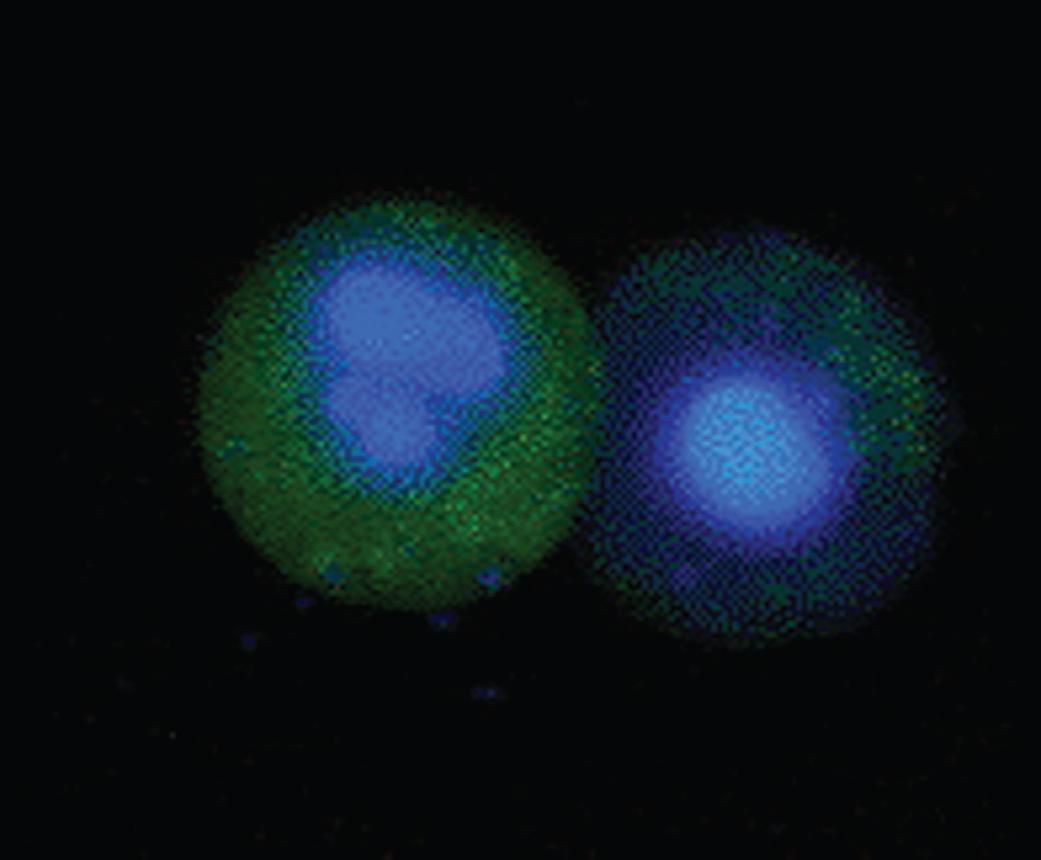
| The FAM FLICA Caspase-1 Kit uses a target sequence (YVAD) sandwiched between a green fluorescent label, carboxyfluorescein (FAM), and a fluoromethylketone (FMK) to make a quick and flexible method to analyze active caspases in apoptotic cells. |
- Reagents In The Kit
-
ICT097: 1 vial of FAM-YVAD-FMK FLICA Reagent - lyophilized
10x Apoptosis Wash Buffer, 15 mL
Fixative, 6 mL
Propidium Iodide, 1 mL
Hoechst 33342, 1 mL. -
ICT098: 4 vials of FAM-YVAD-FMK FLICA Reagent - lyophilized
10x Apoptosis Wash Buffer, 60 mL
Fixative, 6 mL
Propidium Iodide, 1 mL
Hoechst 33342, 1 mL. - Test Principle
-
ICT097: Caspase FLICA kits measure apoptosis by detecting active caspases in whole, living cells. These kits do not work by using antibodies or as an ELISA. Instead, their methodology is based on a unique cell-permeable and non-cytotoxic reagent called the Fluorochrome Inhibitor of Caspases (FLICA). The FLICA reagent contains a caspase inhibitor sequence linked to a green (Carboxyfluorescein, FAM) fluorescent probe.
The Caspase FLICA Kits are suitable for cells in suspension and adherent cells from many species including mammalian, insect and yeast. Different cell types, e.g. HeLa, primary neurons, macrophages and lymphocytes have also been successfully studied with these kits.
This kit can be used with a flow cytometer, fluorescence microscope or a fluorescence plate reader using black microtitre plates. -
ICT098: Caspase FLICA Kits measure apoptosis by detecting active caspases in whole, living cells. These kits do not work by using antibodies or as an ELISA. Instead, their methodology is based on a unique cell-permeable and non-cytotoxic reagent called the Fluorochrome Inhibitor of Caspases (FLICA). The FLICA reagent contains a caspase inhibitor sequence linked to a green (Carboxyfluorescein, FAM) fluorescent probe.
The Caspase FLICA Kits are suitable for cells in suspension and adherent cells from many species including mammalian, insect and yeast. Different cell types, e.g. HeLa, primary neurons, macrophages and lymphocytes have also been successfully studied with these kits.
This kit can be used with a flow cytometer, fluorescence microscope or a fluorescence plate reader using black microtitre plates. - Max Ex/Em
-
Fluorophore Excitation Max (nm) Emission Max (nm) FAM 494 520 - Regulatory
- For research purposes only.
- Guarantee
- Guaranteed until date of expiry. Please see product label.
- Acknowledgements
- FLICA is a trademark of Immunochemistry Technologies, LLC.
Once reconstituted with DMSO, use FLICA reagent immediately, or store at -20°C for 6 months protected from light and thawed no more than twice during that time.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | |||
| Immunofluorescence |
- Instructions For Use
- Instructions for use can be found at www.bio-rad-antibodies.com/uploads/IFU/ICT097.pdf
- Instructions for use can be found at www.bio-rad-antibodies.com/uploads/IFU/ICT098.pdf
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| FAM FLICA™ Caspase-1 Kit | ICT098 | F IF | 100 Tests | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | FAM FLICA™ Caspase-1 Kit | ||||||
References for Caspase-1
-
Inokuchi, T. et al. (2006) Plasma interleukin (IL)-18 (interferon-gamma-inducing factor) and other inflammatory cytokines in patients with gouty arthritis and monosodium urate monohydrate crystal-induced secretion of IL-18.
Cytokine. 33 (1): 21-7. -
Hoegen, T. et al. (2011) The NLRP3 Inflammasome Contributes to Brain Injury in Pneumococcal Meningitis and Is Activated through ATP-Dependent Lysosomal Cathepsin B Release.
J Immunol. 187: 5440-51. -
Wang, Y. et al. (2012) A comparative study of stress-mediated immunological functions with the adjuvanticity of alum.
J Biol Chem. 287 (21): 17152-60. -
Burm, S.M. et al. (2015) Inflammasome-induced IL-1β secretion in microglia is characterized by delayed kinetics and is only partially dependent on inflammatory caspases.
J Neurosci. 35 (2): 678-87. -
Sharma, A.A. et al. (2015) Impaired NLRP3 inflammasome activity during fetal development regulates IL-1β production in human monocytes.
Eur J Immunol. 45 (1): 238-49. -
Wang, Y. et al. (2015) Stress-activated Dendritic Cells (DC) Induce Dual Interleukin (IL)-15- and IL1β-mediated Pathways, Which May Elicit CD4+ Memory T Cells and Interferon (IFN)-stimulated Genes.
J Biol Chem. 290 (25): 15595-609. -
Gabrion, A. et al. (2017) Mammalian target of rapamycin inhibition counterbalances the inflammatory status of immune cells in patients with chronic granulomatous disease.
J Allergy Clin Immunol. 139 (5): 1641-9.e6.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
Always be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up