Complement System Mini-Review
What is the complement system?
The complement system is an ancient part of the innate immune system that acts as the first line of defence in the fight against infection. It is composed of at least 30 proteins, mostly synthesised in the liver, which typically circulate as inactive precursors. Then inactive state of the protease elements is maintained by the presence of several inhibitors. When the complement system is triggered a proteolysis based activation cascade proceeds causing an increased local immune responses and formation of a membrane attack complexes.
There are three biochemical pathways that lead to the activation of the complement system, they require different molecules for initiation but ultimately lead to activation of the same effector molecules:
- Classical complement pathway
- Lectin complement pathway
-
Alternative complement pathway
What are the main functions of the complement system?
- Opsonization – increases phagocytosis by opsonins (C4b and C3b) binding to foreign organisms
- Chemotaxis – attracts macrophages and neutrophils via inflammation by inflammatory mediators; C5a, and to a lesser extent C3a and C4a
- Cell lysis – ruptures membranes due to formation of a membrane attack complex
- Agglutination – causes clustering and binding of pathogens
The pathways
All three pathways converge at the point at which C3 convertase is activated. Pathway complement components are cleaved into two fragments; a larger active serine protease which stays on the pathogen surface (such as C3b, C4b and C5b) and a smaller fragment that is released to act as a soluble mediator (including C3a, C4a, C5a).
The classical complement pathway
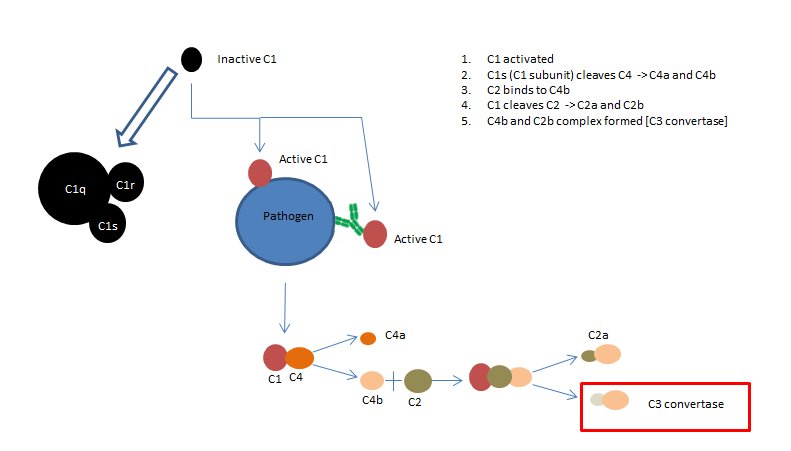
Fig 1. The classical complement pathway
As shown in fig 1. the classical pathway can be initiated in two ways. C1q (part of the C1 complex), either binds to the pathogen surface directly, or binds to the Fc region of an antibody:antigen complex during the adaptive immune response. C1q is a six headed molecule, attached to two molecules C1r and C1s (seen enlarged in fig 1). When two of the C1q heads bind to the pathogen, this causes a conformational change that activates the C1s enzyme. This C1s enzyme goes on to cleave C4 to produce C4b and C4a. C2 binds to C4b (which is bound to the pathogen) allowing it to be cleaved by C1 into C2a and C2b. This results in a C4b and C2b complex, also known as C3 convertase.
The lectin complement pathway
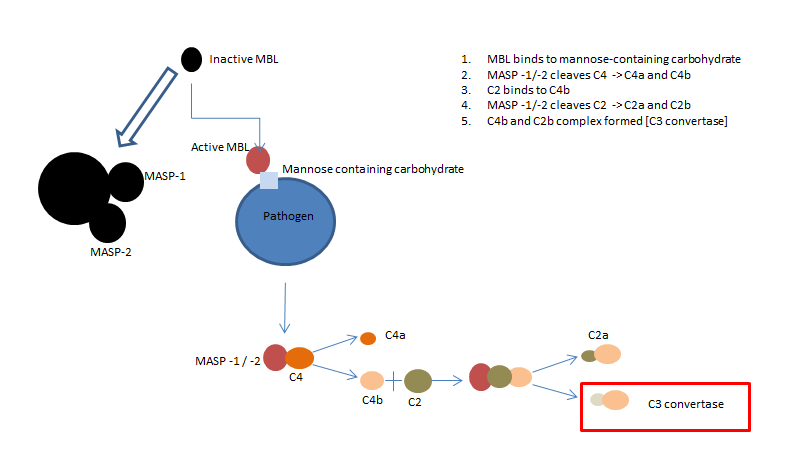
Fig 2. The lectin complement pathway
The lectin complement pathway works in a very similar way to the classical pathway. Instead of the C1 complex, the mannan-binding lectin (MBL) complex binds to a mannose-containing carbohydrate present on the infectious agent. MBL, similar to C1, is a six headed molecule, bound to two protease zymogens called MASP-1 and MASP-2, that cleave C4.
MBL deficiency is associated with an increase in infections during early childhood, indicating the importance of the MBL pathway for host defence (Anders et al. 2001).
The alternative complement pathway
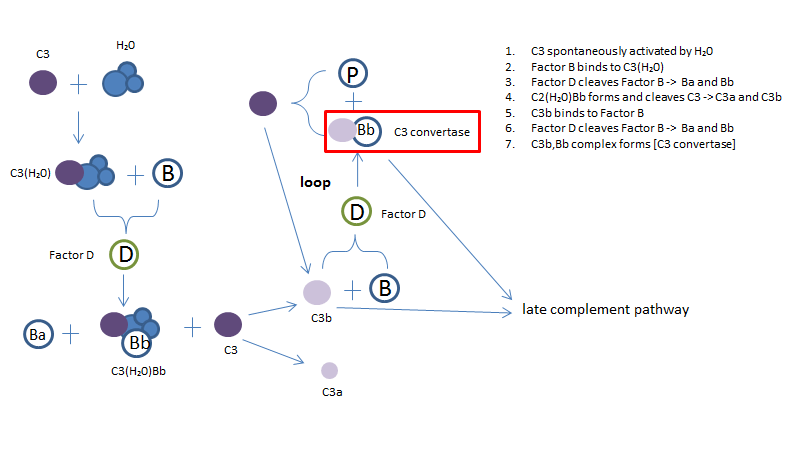
Fig 3. The alternative complement pathway
The alternative pathway, unlike the other two, is not activated by pathogen binding. Instead it is initiated by the spontaneous activation of C3 by water (H2O) via its thio ester bond. This causes a conformational change that binds to Factor B, this C3(H2O)B complex gets cleaved by Factor D to produce complex C3(H2O)Bb (a fluid-phase C3 convertase), which goes on to cleave C3 into C3a and C3b. C3b binds to a pathogen followed by the binding of Factor B, which is then cleaved by Factor D into Ba and Bb. This results with the C3 convertase complex C3bBb. C4bBb then binds to Factor P, which stabilizes the convertase that goes on to cleave C3 to produce C3b. This forms a loops that leads to the creation of more C3b and C3 convertase.
The convergence: beyond C3 convertase
Once the C3 convertase (C4bC2b of the classical and lectin pathways or C3bBb of the alternative pathway) has formed at the pathogen surface, it cleaves C3 to generate C3b and C3a. C3b acts as an opsonin that binds to pathogens marking them as targets for phagocytes; many C3b molecules bind to a pathogen’s cell surface. It also binds to C3 convertase to form C5 convertase, this cleaves C5 to form C5a and C5b. C5a is the most important peptide mediator of inflammation and C5b initiates the ‘late’ events of the complement pathway, resulting in the assembly of a membrane attack complex that creates pores in the cell membrane of the pathogen.
What happens after C5b?
C5b binds to C6, this complex is then bound to C7. The hydrophobic site of C7 then inserts itself into the lipid bilayer of the pathogen. C8 and C9 then bind to the complex and a similar event occurs. C8 is made up two proteins C8ß and C8a. C8ß binds to C5b allowing the hydrophobic site of C8a-. to insert into the bilayer and induce polymerization of 10-16 C9 molecules into a pore-like structure called the membrane attack complex. This is externally hydrophobic allowing it to associate with the lipid bilayer, creating a hydrophilic internal channel that allows the passage of solutes and water into the cell disrupting the cellular integrity of the pathogen.
The role of inflammatory mediators
The small complement fragments (C3a, C4a, and C5a) are known as anaphylotoxins and are responsible for the local inflammatory responses generated following complement acitvation. Their specific effects include smooth muscle contraction, chemotaxis, release of inflammatory mediators and increased vascular permeability.
C5a also acts on neutrophils and monocytes to increase their adherence to vessel walls, their migration towards antigens, and their ability to ingest particles. C5a and C3a mediate their effects via signalling pathways that include G protein coupled receptors.
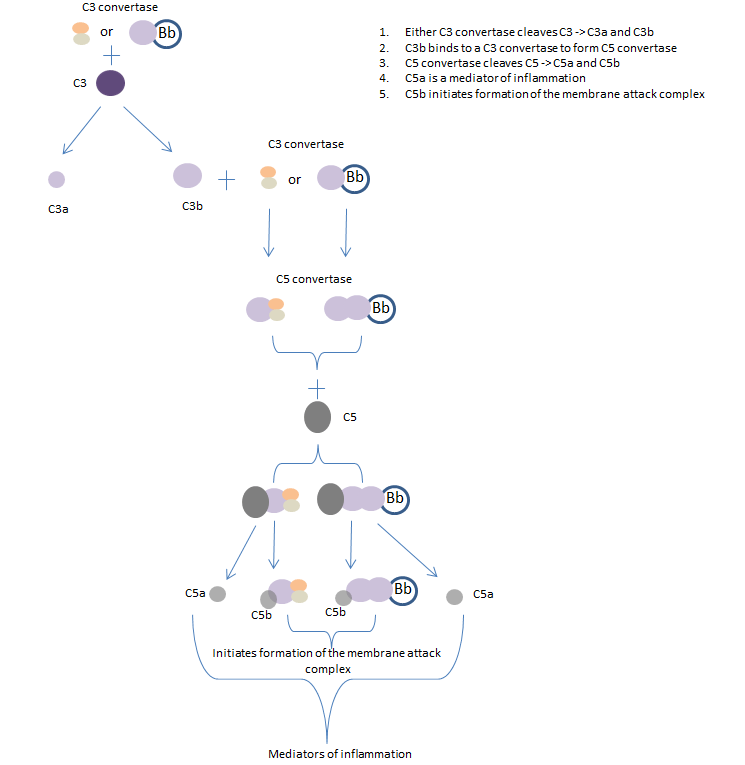
Fig 4. The late complement pathway
Protecting ourselves from the complement pathway…
Under normal conditions the requirement of a foreign pathogen to trigger activation in combination with the transient nature of the activation confines complement appropriately. However, over activation of the complement is associated with disease states such Lupus nephritis. This is often as a result of dysregulation of the systems designed to prevent such uncontrolled activation. These systems include the following:
- C1 inhibitor binds to active C1r: C1s causing it to dissociate from C1q, limiting the ability of C1s to cleave C4 and C2.
- Factor I cleaves C3b into iC3b and then C3dg and cleaves C4b into C4c and C4d, causing them both to inactivate. This occurs when C3b and C4b are bound to cofactor proteins such as CR1 and MCP.
- Factor H in combination with Factor I inactivates C3b
- DAF and CR1 promote the dissociation of active C3 convertase
- Host cell membranes contain an intrinsic protein CD59 that inhibits C9 binding to the C5b678 complex.
The role of complement component C4d
Complement component 4d (c4d) is a protein that acts within the complement system, formed when complement component 4 is cleaved into C4a and C4b; C4b is then subsequently cleaved into its inactive products C4c and C4d by Factor I. The advantage of C4d is that it contains a thiol ester site ensuring it remains covalently bound closeto the site where the C4b was bound. This covalently bound C4d has a much longer half-life than C4b, allowing it to stay anchored to the cell while other molecules get cleared away by the blood stream. This property has established C4d as a stable marker for complement activation and associated transplant rejection. In fact, effective staining of peritubular capillaries has shown it to be a key differentiator between acute humoral rejection (C4d positive) and acute cellular rejection (C4d negative); a distinction that took many years to identify (Collins et al. 1999). Due to this, C4d was made part of the Banff classification in the diagnosis of antibody mediated rejection in allografts (Racusen et al. 2003).
Many therapeutics are under development that target the complement system. C4d can be useful in identifying patients susceptible for those treatments. C4d will likely remain to have a prominent research role in transplantation and autoimmunity.
References
- Anders K et al. (2001) Acute Respiratory Tract Infections and Mannose-Binding Lectin Insufficiency During Early Childhood JAMA. 285:1316-1321.
- Collins BA et al. (1999) Complement Activation in Acute Humoral Renal Allograft Rejection: Diagnostic Significance of C4d Deposits in Peritubular Capillaries. J am Soc Nephorl. 10, 2208-2214.
- Janeway CA Jr et al. (2001) Immunobiology: The Immune System in Health and Disease. 5th edition. New York: Garland Science.
- Neil DA Hubscher SG (2010). Current views on rejection pathology in liver transplantation. Transpl Int 2010; 23: 971–983.
-
Racusen LC et al. (2003) Antibody-mediated rejection criteria—an addition to the Banff 97 classification of renal allograft rejection. Am J Transplant; 3: 708–714.
Further Reading:
- Cohen D et al. (2012) Pros and Cons for C4d as a Biomarker. Kidney Int. 2012;81(7):628-639.
- Sarma JV Ward PA. (2011). The complement system. Cell Tissue Res. 343(1):227-35







