CFDA-SE

CFDA-SE Cell Proliferation Assay Kit
- Product Type
- Accessory Reagent
- Specificity
- CFDA-SE
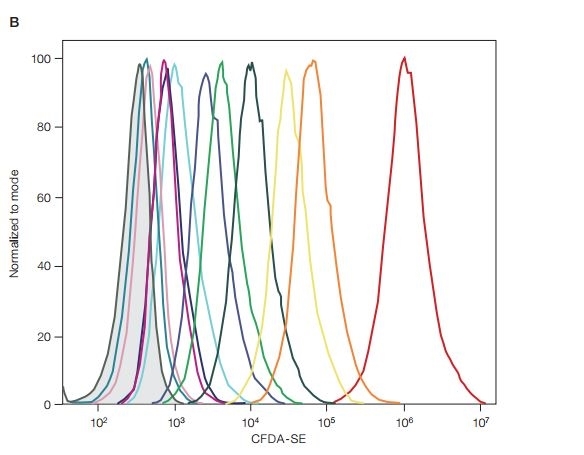
| The CFDA-SE (5[6]-carboxyfluorescein diacetate succinimidyl ester) cell proliferation assay is packaged in 100 μg vials. Simply reconstitute it with DMSO for use and avoid weighing and wasting reagents. CFDA-SE is a cell-permeable reagent that is useful in measuring and tracking cell divisions. Upon entering a live cell, the acetate groups of CFDA-SE are cleaved by intracellular esterase to create the fluorescent carboxyfluorescent carboxyfluorescein succinimidyl ester (CFSE) compound. CFSE reacts with free primary amines to create a stable, covalent bond and is retained in the cytosol of cells. As a cell divides, the fluorescence intensity of CFSE is successively halved with each division, allowing each cell generation to be distinguished. |
- Reagents In The Kit
- Kit includes 5 x 100 μg vials
- Regulatory
- For research purposes only
- Guarantee
- Guaranteed until date of expiry. Please see product label.
This product is photosensitive and should be protected from light
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/40 | 1/400 |
- Instructions For Use
- Important: Thaw all components prior to use.
Note: The following protocol is a guideline and it should be modified for each experiment as needed.
1. Prepare a 200 μM stock solution by adding 892.5 μl of DMSO to a CFDA-SE vial and mix by vortexing.
2. Create a working solution (5-0.5 μM) by diluting the CFDA-SE stock solution from step 1 with your buffer of choice at pH 7.
3. Re-suspend 1 x 106 cells of interest in 500 μl of the working solution.
4. Incubate the cells for 5-20 min at room temperature. Protect from light.
5. Centrifuge the sample and remove the supernatant.
6. Wash the pellet with 3 ml of your buffer of choice.
7. Re-suspend the cells in 500 μl of fresh, pre-warmed culture media.
8. Remove 200 μl to analyze for time zero.
9. Place the remaining cells in the appropriate conditions for cell proliferation.
10. Harvest the cells and stain them for the other markers if desired.
11. Analyze or sort the cells using a flow cytometer or S3e cell sorter with a 488 nm laser.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
Always be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up