Annexin V Kits: For Apoptosis Detection

- On This Page
- Apoptosis detection by Flow Cytometry
- Annexin V kit benefits
- Our Annexin V kit range
Apoptosis Detection by Flow Cytometry
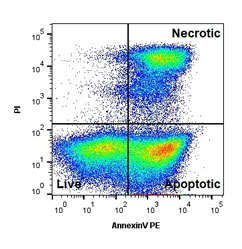
Apoptosis, or programmed cell death, is an essential process by which multi-cellular organisms ensure that critically damaged or mutated cells are removed. The process is regulated by a complex interplay of proteins and, until reaching “the point of no return”, can be halted or reversed at any point.
Early apoptotic cells can be detected with the help of annexin V; a protein encoded by the ANXA5 gene, which acts as an anticoagulant. Annexin V, as other members of the annexin protein family, binds to specific membrane proteins. During the early stages of apoptosis the plasma membrane is rearranged, which results in changes to the cell membrane’s phospholipid asymmetry. One of the effects of these rearrangements is the display of phosphatidylserine (PS) at the outer plasma membrane. In healthy, quiescent cells PS is exclusively located at the inner membrane and therefore the exposure of PS is an indication of early apoptosis (Fadeel and Xue, 2009). Annexin V has a high binding affinity for PS in the presence of Ca2+ and is therefore a common tool for detecting apoptotic cells.
Annexin V Kit Benefits
- Offered in a variety of sizes, ranging from 20/50 tests trial sizes to a 300 tests size for frequent users
- Enable the use of the kits in multi-color flow cytometry experiments; conjugates available include Biotin, FITC, PE and APC
- Include the viability dye propidium iodide enabling distinction between apoptotic/secondary necrotic and apoptotic cells
Labeled annexin V, included in the kits, has been conjugated such that the native phospholipid binding properties of annexin V remain unaltered.
Our Annexin V Kit Range
| Description | Target | Format | Clone | Applications | Citations | Code |
|---|
Reference
- Fadeel B and Xue D (2009). The ins and outs of phospholipid asymmetry in the plasma membrane: roles in health and disease.Crit. Rev. Biochem. Mol. Biol. 44:264-277.


